Chapter 0: Q50P (page 1)
Write the reaction of phosphoric acid and the reaction of disodium oxalate.
Short Answer
The reaction of phosphoric acid:
The reaction of disodium oxalate:
Learning Materials
Features
Discover
Chapter 0: Q50P (page 1)
Write the reaction of phosphoric acid and the reaction of disodium oxalate.
The reaction of phosphoric acid:
The reaction of disodium oxalate:
All the tools & learning materials you need for study success - in one app.
Get started for free
The figure shows reversed-phase retention data for three compounds.
(a) Identify whether compounds A, B, and C are weak acids or bases. For each compound, what isand the retention factor of the fully protonated form?
(b) Over what pHrange would a method be least rugged with regard to retention of component C?
(c) Each different symbol in the plot indicates a different buffer (circle pH2.48phosphate; plus pH5.01acetate; and so on). Why are different buffers used for this experiment?
When you flip a coin, the probability of its landing on each side isin Equations 28-2 and 28-3. If you flip it times, the expected number of heads equals the expected number of tailsThe expected standard deviation for flips is. From Table 4-1, we expect thatof the results will lie withinand of the results will lie within.
(a) Find the expected standard deviation for the number of heads incoin flips.
(b) By interpolation in Table 4-1, find the value of that includesof the area of the Gaussian curve. We expect thatof the results will lie within this number of standard deviations from the mean.
(c) If you repeat thecoin flips many times, what is the expected range for the number of heads that includesof the results? (For example, your answer might be, "The rangetowill be observedof the time.")
Protein and carbohydrates provide, whereas fat gives . (Remember that 1 Calorie , with a capital C, is really .) The weight percentages of these components in some foods are
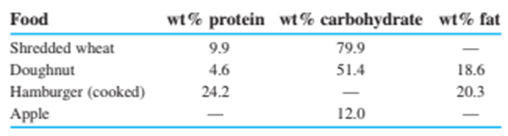
Calculate the number of calories per gram and calories per ounce in each of these foods. (Use Table 1-4to convert grams into ounces, remembering that there are 16ounces in 1pound.)
Succinic acid dissociates in two steps:

Is it possible to precipitate of role="math" localid="1663573455203" by adding oxalate without precipitating ?
role="math" localid="1663574065069"
What do you think about this solution?
We value your feedback to improve our textbook solutions.