Chapter 0: Q6-I E (page 1)
Succinic acid dissociates in two steps:

Short Answer
Base dissociation constant for given reaction is,
Learning Materials
Features
Discover
Chapter 0: Q6-I E (page 1)
Succinic acid dissociates in two steps:

Base dissociation constant for given reaction is,
All the tools & learning materials you need for study success - in one app.
Get started for free
How many milliliters of 2.15% alcoholic dimethylglyoxime should be used to provide a 50.0% excess for Reaction 27-7 with 0.998 4 g of steel containing 2.07 wt% Ni? Assume that the density of the dimethylglyoxime solution is 0.790 g/mL.
Theof microscopic vesicles (compartments) in living cells can be estimated by infusing an indicator (HIn) into the compartment and measuring the quotientfrom the spectrum of the indicator inside the vesicle. Explain how this tells us the.
A solid mixture weighing 0.05485 gcontained only ferrous ammonium sulphate and ferrous chloride. The sample was dissolved in, and the required 13.39 mL oflocalid="1663577431957" for complete oxidation to localid="1663577453691" . Calculate the weight percent of Cl in the original sample. If you need refreshing, see Section 7-2 for an example of a titration of a mixture.
localid="1663574328546"
Ferrous ammonium sulfate Ferrous chloride
FM 392.13
FM 234.84
Describe how you would prepare exactlyof picolinate buffer, . Possible starting materials are pure picolinic acid (pyridine-2-carboxylic acid, FM 123.11), 1.0 M HCl, and . Approximately how many milliliters of the orwill be required?
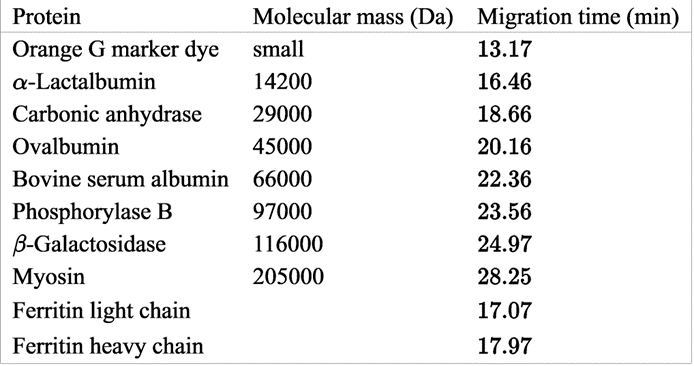
Molecular mass by sodium dodecylsulfate–gel electrophoresis. Ferritin is a hollow iron-storage protein 74 consisting of 24 subunits that are a variable mixture of heavy (H) and light (L) chains,arranged in octahedral symmetry. The hollow center, with a diameter of 8 nm, can hold up to 4 500 iron atoms in the approximate form of the mineral ferrihydrite Iron(II) enters the protein through eight pores located on the threefold symmetry axes of the octahedron. Oxidation to Fe(III) occurs at catalytic sites on the H chains. Other sites on the inside of the L chains appear to nucleate the crystallization of ferrihydrite. Migration times for protein standards and the ferritin subunits are given in the table. Prepare a graph of log(molecular mass) versus 1/(relative migration time), where relative migration time 5 (migration time)/(migration time of marker dye). Compute the molecular mass of the ferritin light and heavy chains. The masses of the chains, computed from amino acid sequences, are 19 766 and 21 099 Da.
What do you think about this solution?
We value your feedback to improve our textbook solutions.