Chapter 0: Q31 P (page 1)
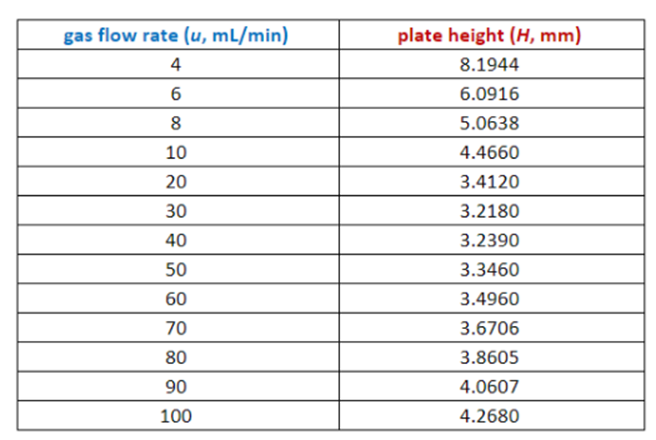
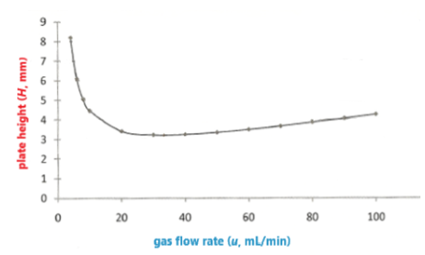
The efficiency of a gas chromatography column is measured by a parameter called plate height (H, m m) , which is related to the gas flow rate (u, mL/min) by the van Deemter equation: H = A + B / u + Cu , where A , B , and C are constants. Prepare a spreadsheet with a graph showing values of as a function of u for u =4,6,8,10,20,20,40,50,60,70,80,90 and 10 mL/min. Use the values A = 1.65 m m , B = 25.8 m m .m L/min, and C = 0.023 6 m m.min/mL.
Short Answer
The spreadsheet and the graph are in step 2 and 3.