Chapter 0: Q11P (page 1)
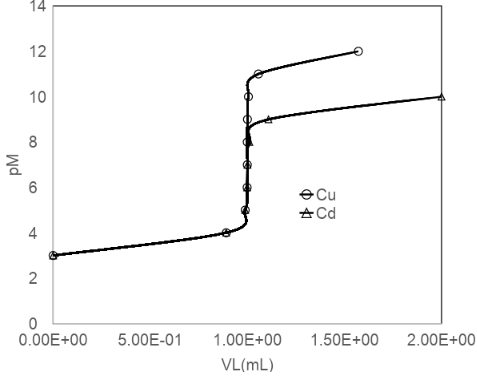
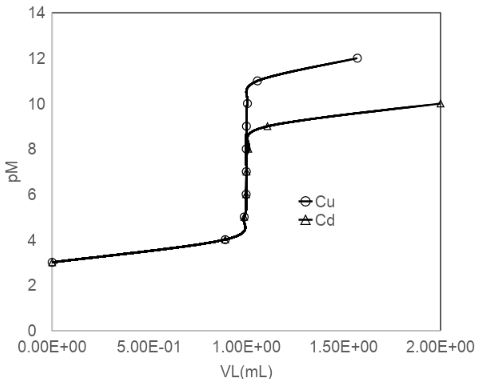
Titration of metal ion with EDTA. Use Equation 12-11 to compute curves (pM versus mL of EDTA added) for the titration of 10.00 mL of 1.00 mM M2+ (= Cd2+ or Cu2+) with 10.0 mM EDTA at pH 5.00. Plot both curves on one graph.
Short Answer
The curves for both the metals Cd and Cu are shown below