Chapter 0: Q6-DE (page 1)
Which will be more soluble (moles of metal dissolved per litre of solution), ? Give an example of a chemical reaction that might occur that would reverse the predicted solubilities.
Short Answer
is found to be more soluble.
Learning Materials
Features
Discover
Chapter 0: Q6-DE (page 1)
Which will be more soluble (moles of metal dissolved per litre of solution), ? Give an example of a chemical reaction that might occur that would reverse the predicted solubilities.
is found to be more soluble.
All the tools & learning materials you need for study success - in one app.
Get started for free
25. Write a mass balance for a solution ofif the species
areandrole="math" localid="1663402048017" .
When you flip a coin, the probability of its landing on each side isin Equations 28-2 and 28-3. If you flip it times, the expected number of heads equals the expected number of tailsThe expected standard deviation for flips is. From Table 4-1, we expect thatof the results will lie withinand of the results will lie within.
(a) Find the expected standard deviation for the number of heads incoin flips.
(b) By interpolation in Table 4-1, find the value of that includesof the area of the Gaussian curve. We expect thatof the results will lie within this number of standard deviations from the mean.
(c) If you repeat thecoin flips many times, what is the expected range for the number of heads that includesof the results? (For example, your answer might be, "The rangetowill be observedof the time.")
List the steps in a chemical analysis.
The iodide content of a commercial mineral water was measured by two methods that produced wildly different results. Method A found milligrams of per literand method B found 0.009mg/L. When was added to the water, the content found by method A increased each time that morewas added, but results from method B were unchanged. Which of the Terms to Understand describes what is occurring in these measurements? Explain your answer. Which result is more reliable?
What is the purpose of the trap in Figure 2-17 and the watch glass in Figure 2-20?
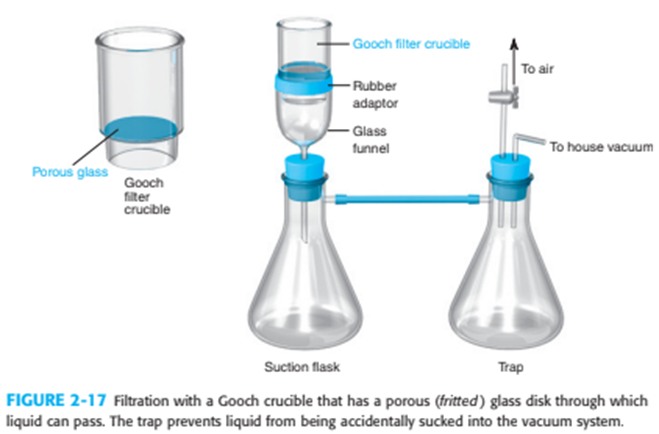
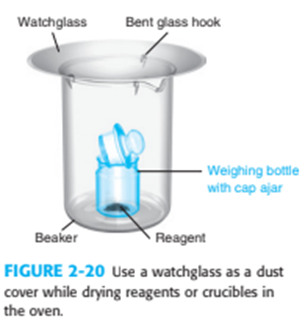
What do you think about this solution?
We value your feedback to improve our textbook solutions.