Chapter 0: Q1TY (page 1)
25-1 What should be the volume flow rate and injected volume for a 1.5-mm-diameter column?
Short Answer
The injected volume in the given column is calculated as.
Learning Materials
Features
Discover
Chapter 0: Q1TY (page 1)
25-1 What should be the volume flow rate and injected volume for a 1.5-mm-diameter column?
The injected volume in the given column is calculated as.
All the tools & learning materials you need for study success - in one app.
Get started for free
(a) What is the pH at the equivalence point when 0.030 0 M NaF is titrated with 0.060 0 M HClO4?
(b) Why would an indicator endpoint probably not be useful in this titration?
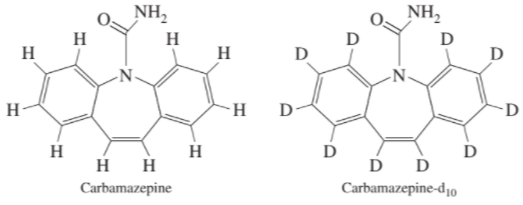
Correcting for matrix effects with an internal standard. The appearance of pharmaceuticals in municipal wastewater (sewage) is an increasing problem that is likely to have adverse effects on our drinking water supply. Sewage is a complex matrix. The drug carbamazepine can be measured at low levels in sewage by liquid chromatography with mass spectral detection. When carbamazepine was spiked into sewage at a concentration of
chromatographic analysis gave an apparent spike recovery of Deuterium (D) is the hydrogen isotope . Deuterated carbamazepine can be used as an internal standard for carbamazepine. The deuterated compound has the same retention time as undertreated material in chromatography, but is distinguished by its higher mass in the mass spectrum. When deuterated carbamazepine was used as an internal standard for the analysis, the apparent recovery was Explain how the internal standard is used in this analysis and rationalize why it works so well to correct for matrix effects.
Describe how you would prepare exactlyof picolinate buffer, . Possible starting materials are pure picolinic acid (pyridine-2-carboxylic acid, FM 123.11), 1.0 M HCl, and . Approximately how many milliliters of the orwill be required?
Calculate the formula mass of. What is the molarity of in a solution containing 1.2gof in a volume of 50 mL? How many grams of are in 50 mLof ?
What measures can be taken to decrease the relative supersaturation during a precipitation?
What do you think about this solution?
We value your feedback to improve our textbook solutions.