Chapter 31: Q22E (page 1054)
Draw the structure of Kedol, a polyester prepared by heating dimethyl 1,4- benzene dicarboxylate with 1,4- bis (hydroxymethyl) cyclohexane.
Short Answer
The answer is,
Learning Materials
Features
Discover
Chapter 31: Q22E (page 1054)
Draw the structure of Kedol, a polyester prepared by heating dimethyl 1,4- benzene dicarboxylate with 1,4- bis (hydroxymethyl) cyclohexane.
The answer is,
All the tools & learning materials you need for study success - in one app.
Get started for free
1,3-Cyclopentadiene undergoes thermal polymerization to yield a polymer that has no double bonds in the chain. Upon strong heating, the polymer breaks down to generate cyclopentadiene. Propose a structure of the polymer.
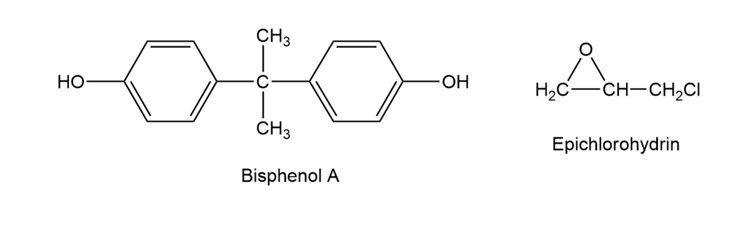
Epoxy adhesives are cross-linked resins prepared in two steps. The first step involved the SN2 reaction of the disodium salt of bisphenol A with epichlorohydrin to form a low molecular weight prepolymer. This polymer is then cured into a cross-linked resin by treatment with a triamine such as
Poly(ethylene terephthalate), or PET, is a polyester used to make soft-drink bottles. It is prepared by reaction of ethylene glycol with 1,4-benzenedicarboxylic acid (terephthalic acid). Draw the structure of PET.
Poly (vinyl butyral) is used as the plastic laminate in the preparation of automobile windshield safety glass. How would you synthesize this polymer?
Nylon 10, 10 is an extremely tough, strong polymer used to make reinforcing rods for concrete. Draw a segment of nylon 10, 10, and show its monomer units.
What do you think about this solution?
We value your feedback to improve our textbook solutions.