Chapter 13: Q13-37E (page 419)
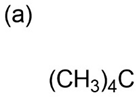
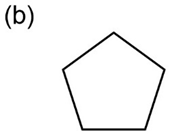
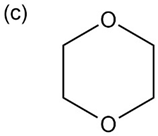
Propose structures for compounds with the following formulas that show only one peak in their NMR spectra:
a.
b.
c.
Short Answer
Learning Materials
Features
Discover
Chapter 13: Q13-37E (page 419)
Propose structures for compounds with the following formulas that show only one peak in their NMR spectra:
a.
b.
c.



All the tools & learning materials you need for study success - in one app.
Get started for free
How could you use 1H NMR to determine the regiochemistry of electrophilic addition to alkenes? For example, does addition of HCl to 1-methylcyclohexene yield 1-chloro-1-methylcyclohexane or 1-chloro-2-methylcyclohexane?
Long-range coupling between protons more than two carbon atoms apart is sometimes observed when p bonds intervene. An example is found in 1-methoxy-1-buten-3-yne. Not only does the acetylenic proton, , couple with the vinylic proton , it also couples with the vinylic proton , four carbon atoms away. The data are:
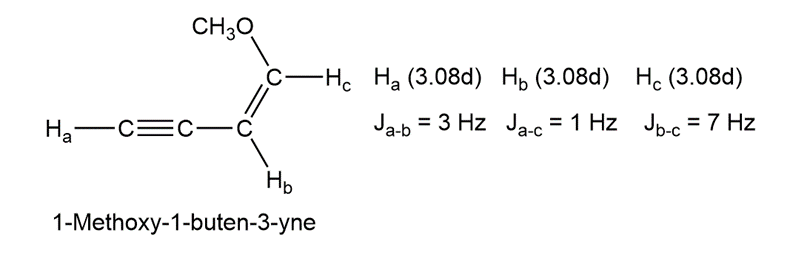
Construct tree diagrams that account for the observed splitting patterns of ,, and .
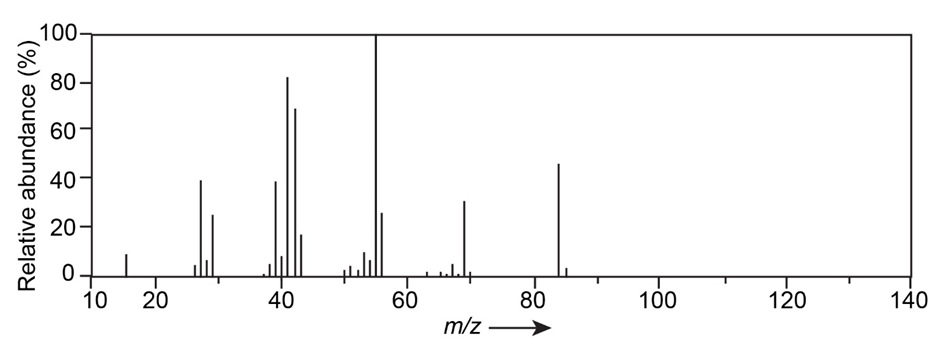
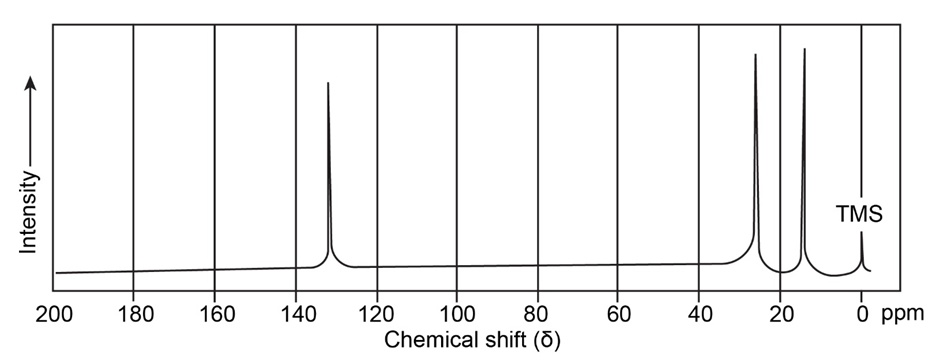
Question: The mass spectrum and NMR spectrum of a hydrocarbon are shown. Propose a structure for this hydrocarbon, and explain the spectral data.
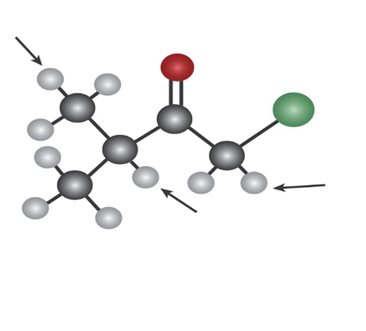
Into how many peaks would you expect the 1H NMR signals of the indicated protons to be split? (Green=Cl.)
a)
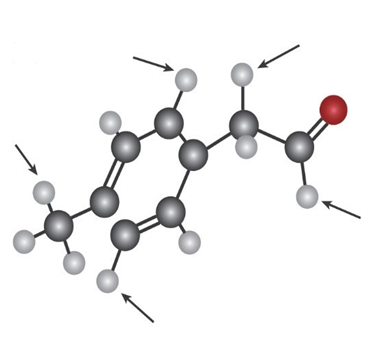
b)
The following NMR peaks were recorded on a spectrometer operating at
200 MHz. Convert each into O1units.
(a) CH3CI; 1454Hz
(b) CHCI3; 610Hz
(c) CH3OH; 693Hz
(d) CH2CI2; 1060Hz
What do you think about this solution?
We value your feedback to improve our textbook solutions.