Chapter 13: Q13-25E (page 419)
How many absorptions would you expect the following compound tohave in its 1H and 13C NMR spectra?
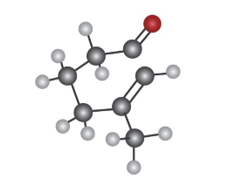
Short Answer
Learning Materials
Features
Discover
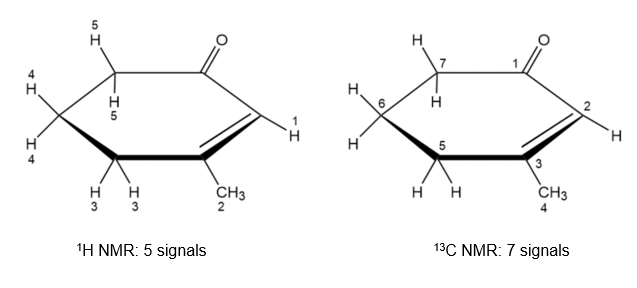
Chapter 13: Q13-25E (page 419)
How many absorptions would you expect the following compound tohave in its 1H and 13C NMR spectra?


All the tools & learning materials you need for study success - in one app.
Get started for free
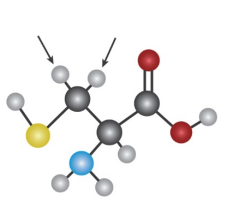
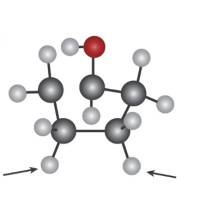
Question: Identify the indicated protons in the following molecules as unrelated,
homotopic, enantiotopic, or diastereotopic.
a)
b)
Propose a structure for an aromatic hydrocarbon, C11H16, that has the following 13C NMR spectral data:
Broadband decoupled: 29.5, 31.8, 50.2, 125.5, 127.5, 130.3, 139.8
DEPT-90: 125.5, 127.5, 130.3
DEPT-135: positive peaks at 29.5, 125.5, 127.5, 130.3d;negative peak at
50.2
We saw in Section 9-3 that addition of H-Br to a terminal alkyne leads to the Markovnikov addition product, with the Br bonding to the more highly substituted carbon. How could you use 13C NMR to identify the product of theaddition of 1 equivalent of H-Br to 1-hexyne?
Assign a chemical shift to each carbon in 6-methyl-5-hepten-2-ol.
Predict the splitting pattern for each kind of hydrogen in the following molecules:
(a)
(b)
(c) trans-2-Butene
What do you think about this solution?
We value your feedback to improve our textbook solutions.