Chapter 12: Q12-41E (page 354)
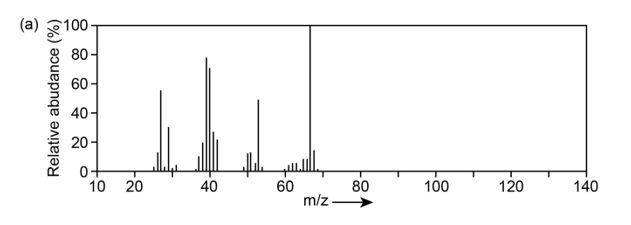
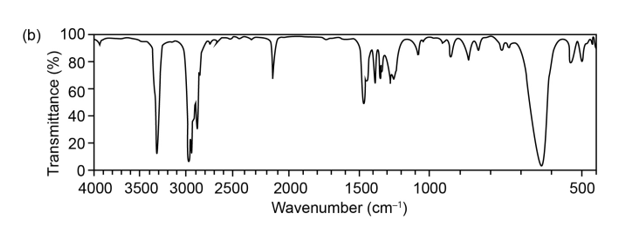
The mass spectrum (a) and the infrared spectrum (b) of an unknownhydrocarbon are shown. Propose as many structures as you can.
Short Answer
Learning Materials
Features
Discover
Chapter 12: Q12-41E (page 354)
The mass spectrum (a) and the infrared spectrum (b) of an unknownhydrocarbon are shown. Propose as many structures as you can.



All the tools & learning materials you need for study success - in one app.
Get started for free
Write molecular formulas for compounds that show the following molecularions in their high-resolution mass spectra, assuming that C, H, N,and O might be present. The exact atomic masses are: 1.00783(1H), 12.00000(12C) , 14.00307 (14N) , 15.99491(16O) .
(a) M+ = 98.0844
(b) M+= 123.0320
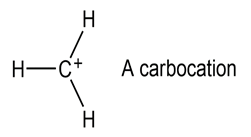
Most stable organic species have tetravalent carbon atoms, but species with trivalent carbon atoms also exist. Carbocations are one such class of compounds.
(a) How many valence electrons does the positively charged carbon atom have?
(b) What hybridization do you expect this carbon atom to have?
(c) What geometry is the carbocation likely to have?
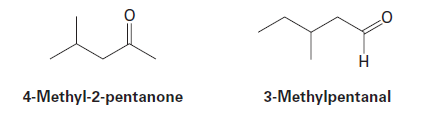
Question: 4-Methyl-2-pentanone and 3-methylpentanal are isomers. Explain how you could tell them apart, both by mass spectrometry and by infrared spectroscopy.
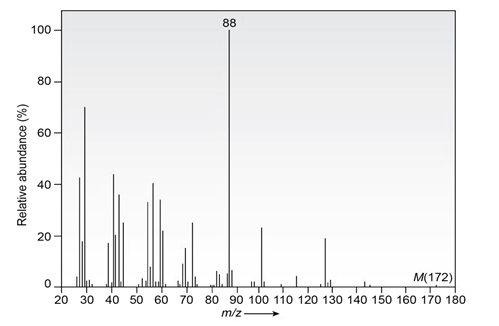
Question: The infrared spectrum of the compound with the mass spectrum shown below lacks any significant absorption above . There is a prominent peak near and another strong peak near .Propose a structure consistent with the data.
Draw structures corresponding to the following IUPAC names:
a)cis-1,2-Cyclohexanedicarboxylic acid
b) Hepatanedioic acid
c) 2-Hexen-4-ynoic acid
d) 4-Ethyl-2-propyloctanoic acid
e) 3-chlorophthalic acid
f) Triphenylacetic acid
g) 2-Cyclobutenecarbonitrile
h) m-Benzoylbenzonitrile
What do you think about this solution?
We value your feedback to improve our textbook solutions.