Chapter 1: Q44E-a (page 27)
Tell the number of hydrogens bonded to each carbon atom in the following substances, and give the molecular formula of each:
(a)
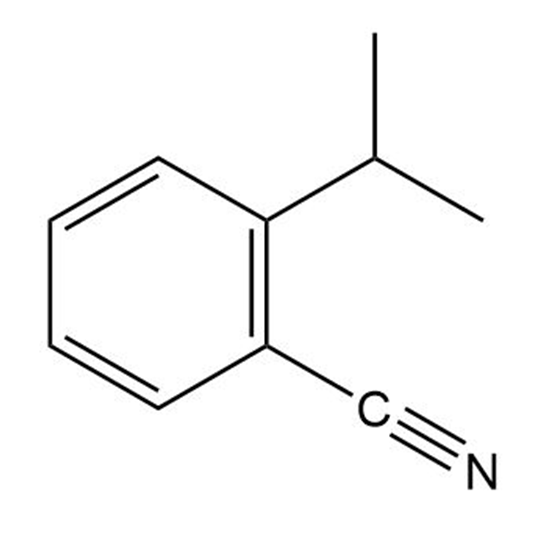
(b)
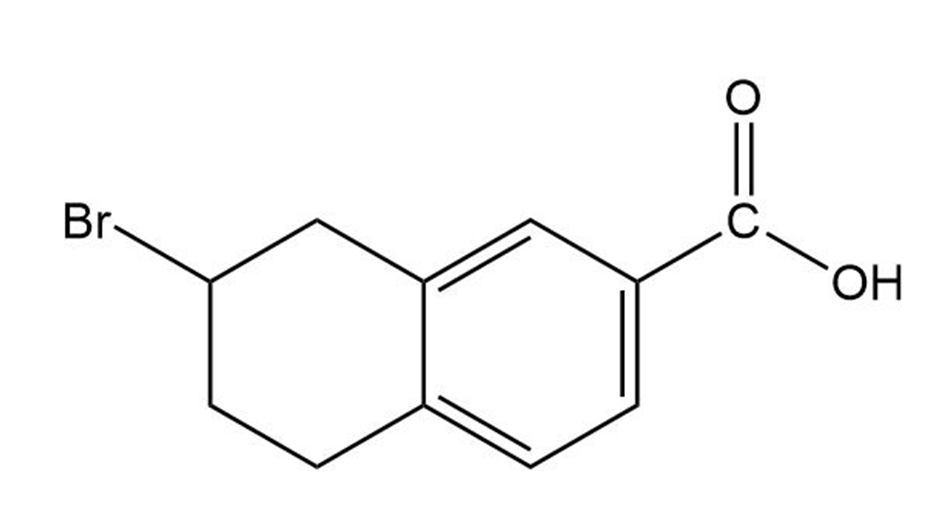
(c)
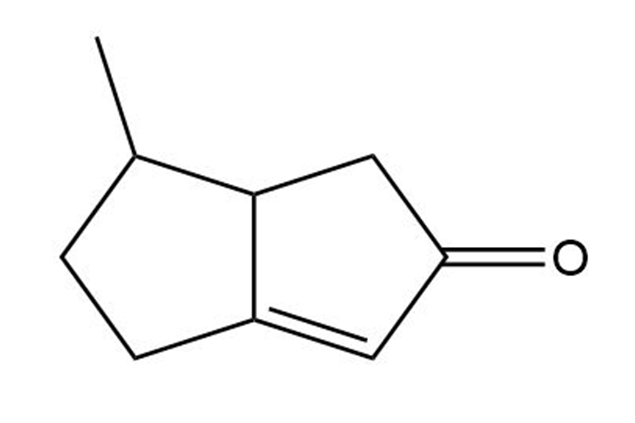
Short Answer
(a)
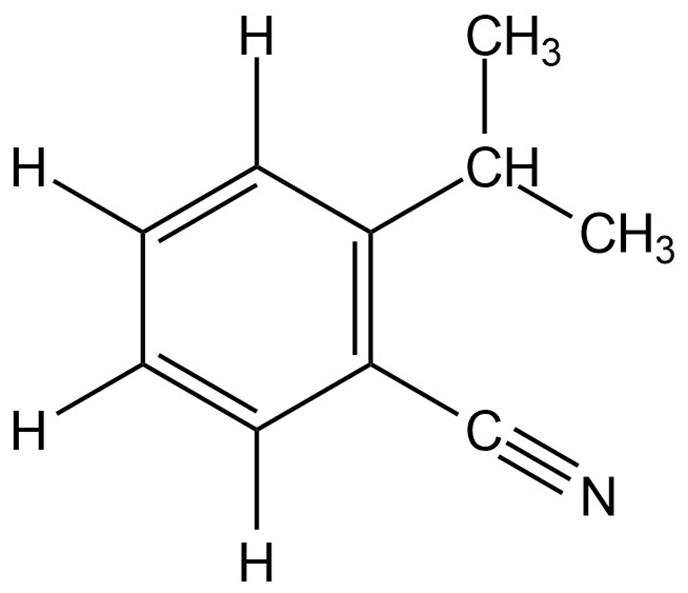
Number of hydrogen atoms bonded to carbon
Learning Materials
Features
Discover
Chapter 1: Q44E-a (page 27)
Tell the number of hydrogens bonded to each carbon atom in the following substances, and give the molecular formula of each:
(a)

(b)

(c)

(a)

Number of hydrogen atoms bonded to carbon
All the tools & learning materials you need for study success - in one app.
Get started for free
Oxaloacetic acid, an important intermediate in food metabolism, has the formulaand contains threebonds and twobonds. Propose two possible structures.
Divalent carbon species called carbenes are capable of fleeting existence. For example, methylene, :CH2, is the simplest carbene. The two unshared electrons in methylene can be either paired in a single orbital or unpaired in different orbitals. Predict the type of hybridization you expect carbon to adopt in singlet (spin-paired) methylene and triplet (spin-unpaired) methylene. Draw a picture of each, and identify the valence orbitals on carbon.
Draw structures for the following molecules, showing lone pairs:
(a) Acrylonitrile, C3H3N, which contains a carbon-carbon double bond and a carbon-nitrogen triple bond
(b) Ethyl methyl ether,C3H8O, which contains an oxygen atom bonded to two carbons
(c) Butane,C4H10, which contains a chain of four carbon atoms
(d) Cyclohexene,C6H10, which contains a ring of six carbon atoms and one carbon-carbon double bond
Allene, is somewhat unusual in that it has two adjacent double bonds. Draw a picture showing the orbitals involved in the s and p bonds of allene. Is the central carbon atomsp2orsp-hybridized? What about the hybridization of the terminal carbons? What shape do you predict for allene?
Among the most common over-the-counter drugs you might find in a medicine cabinet are mild pain relievers such ibuprofen (Advil, Motrin), naproxen (Aleve), and acetaminophen (Tylenol).
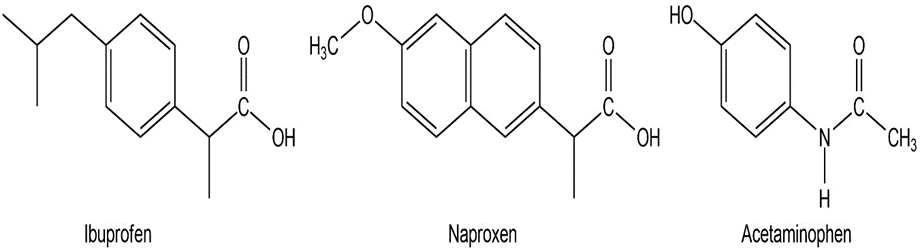
(a) How many sp3-hybridized carbons does each molecule have?
(b) How many sp2-hybridized carbons does each molecule have?
(c) Can you spot any similarities in their structures?
What do you think about this solution?
We value your feedback to improve our textbook solutions.