Chapter 1: Q1-1-43E c (page 27)
Tell the number of hydrogens bonded to each carbon atom in the following substances, and give the molecular formula of each:
(c)
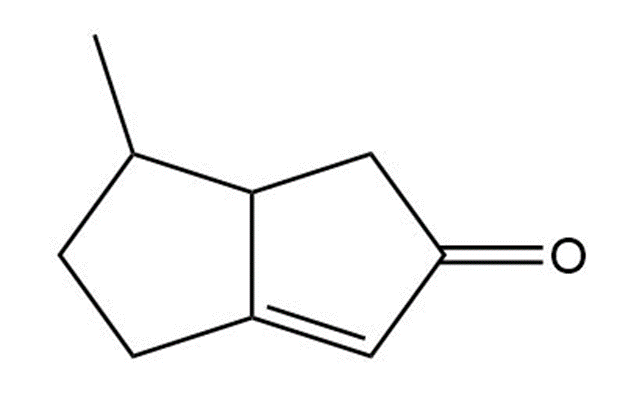
Short Answer
Number of hydrogen atoms bonded to carbon is 13
Learning Materials
Features
Discover
Chapter 1: Q1-1-43E c (page 27)
Tell the number of hydrogens bonded to each carbon atom in the following substances, and give the molecular formula of each:
(c)


Number of hydrogen atoms bonded to carbon is 13
All the tools & learning materials you need for study success - in one app.
Get started for free
Potassium methoxide,,KOCH3 contains both covalent and ionic bonds. Which do you think is which?
Tell the number of hydrogens bonded to each carbon atom in
(a) the anti-influenza agent oseltamivir, marketed as Tamiflu, and
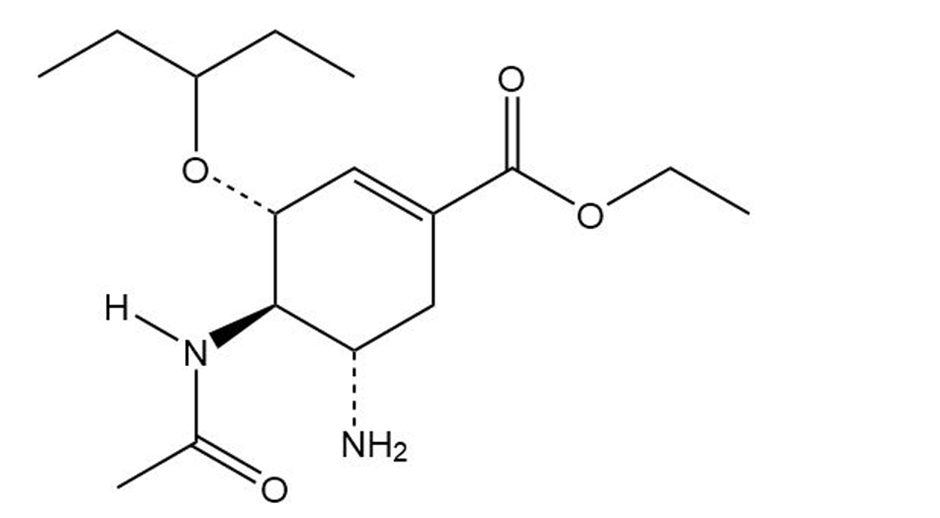
Oseltamivir
(Tamiflu)
(b) the platelet aggregation inhibitor clopidogrel, marketed as Plavix. Give the molecular formula of each
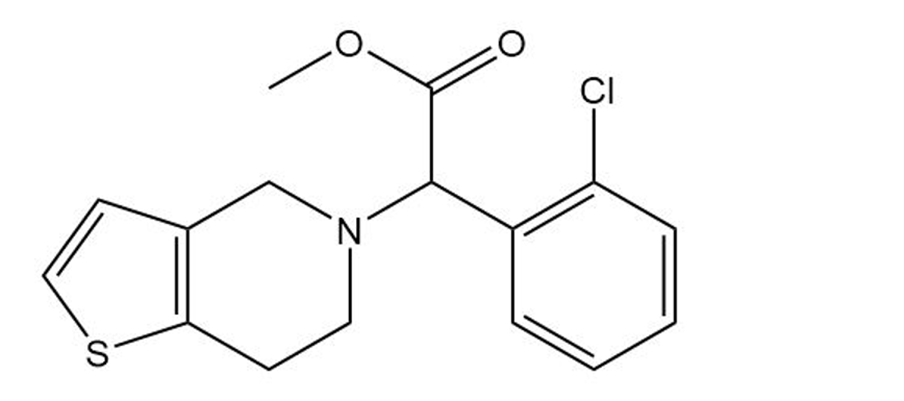
Clopidogrel (Plavix)
Tell the number of hydrogens bonded to each carbon atom in the following substances, and give the molecular formula of each:
(a)
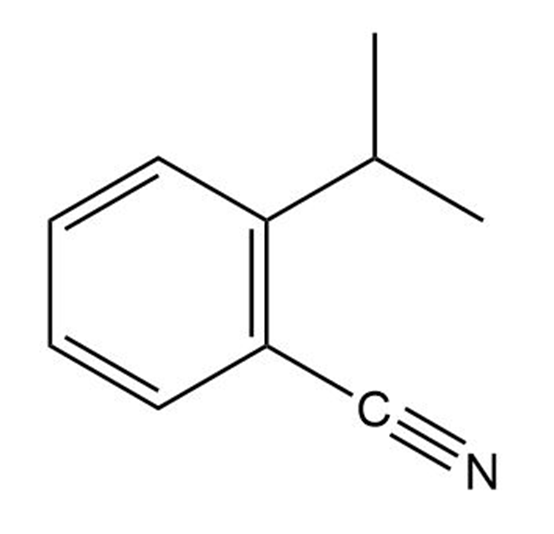
(b)
Quetiapine(Seroquel)
(c)
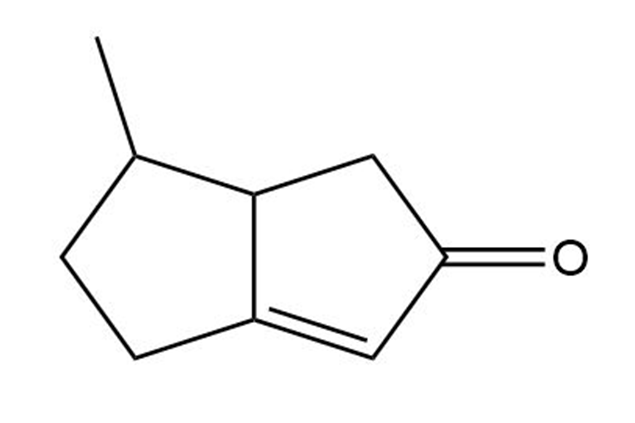
There are two different substances with the formula C3H6. Draw both, and tell how they differ.
What are likely formulas for the following molecules?
(a) (b) (c) (d)
What do you think about this solution?
We value your feedback to improve our textbook solutions.