Chapter 5: Q56 E_a. (page 148)
Identify the indicated faces in the following molecules as Re or Si:
(a)
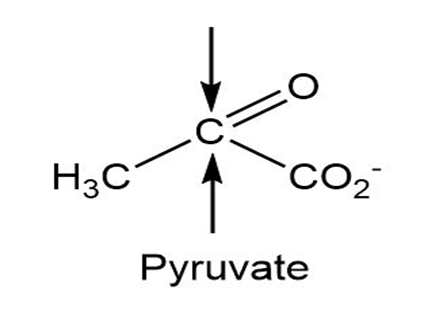
(b)
Short Answer
(a)
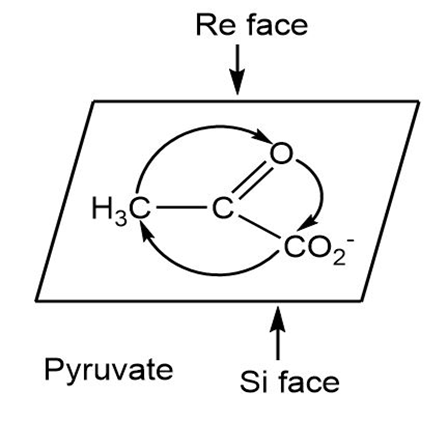
Learning Materials
Features
Discover
Chapter 5: Q56 E_a. (page 148)
Identify the indicated faces in the following molecules as Re or Si:
(a)

(b)

(a)

All the tools & learning materials you need for study success - in one app.
Get started for free
Draw tetrahedral representations of the following molecules: S-2-chlorobutane
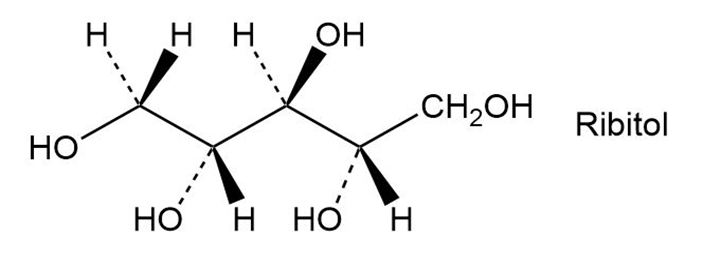
On reaction with hydrogen gas by a platinum catalyst, ribose (Problem 5-54) is converted into ribitol. Is ribitol optically active or inactive? Explain.
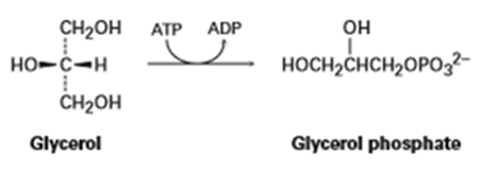
The first step in the metabolism of glycerol, formed by digestion of fats, is phosphorylation of the pro-R group by reaction with adenosine triphosphate (ATP) to give the corresponding glycerol phosphate plus adenosine diphosphate (ADP). Show the stereochemistry of the product.
Draw both cis- and trans-1,3-dimethyl cyclohexane in their more stable chair conformations.
(a) How many stereoisomers are there of cis-1,3-dimethyl cyclohexane, and how many of trans-1,3-dimethyl cyclohexane?
(b) Are any of the structures chiral?
(c) What are the stereochemical relationships among the various stereoisomers of 1,3-dimethyl cyclohexane?
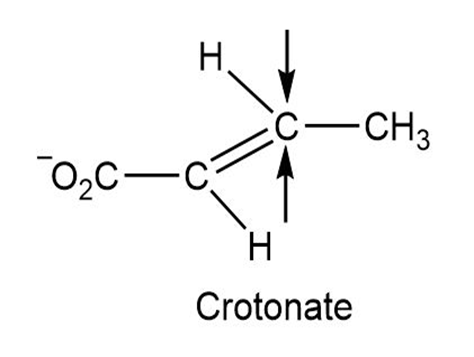
One of the steps in fat metabolism is the hydration of crotonate to yield 3-hydroxybutyrate. This reaction occurs by addition of –OH to the Si face at C3, followed by protonation at C2, also from the Si face. Draw the product of the reaction, showing the stereochemistry of each step.
What do you think about this solution?
We value your feedback to improve our textbook solutions.