Chapter 11: Q11-76E (page 350)
The reaction of 1-chlorooctane with CH3CO2 2 to give octyl acetate is greatly accelerated by adding a small quantity of iodide ion. Explain
Short Answer
 Formation of the desired product
Formation of the desired product
Learning Materials
Features
Discover
Chapter 11: Q11-76E (page 350)
The reaction of 1-chlorooctane with CH3CO2 2 to give octyl acetate is greatly accelerated by adding a small quantity of iodide ion. Explain
 Formation of the desired product
Formation of the desired product
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: (R)-2-Bromooctane undergoes racemization to give ()-2-bromooctane when treated with NaBr in dimethyl sulfoxide. Explain.
Compound X is optically inactive and has the formula. On treatment with strong base, X gives hydrocarbon Y,. Compound Y absorbs 2 equivalents of hydrogen when reduced over a palladium catalyst and reacts with ozone to give two fragments. One fragment, Z, is an aldehyde with formula. The other fragment is glyoxal,. Write the reactions involved, and suggest structures for X, Y, and Z. What is the stereochemistry of X?
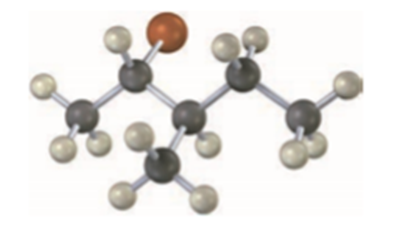
What stereochemistry do you expect for the trisubstituted alkene obtained by E2 elimination of the following alkyl halide on treatment with KOH? (Reddish brown = Br.)
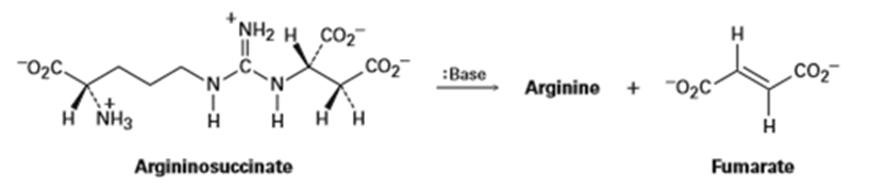
One step in the urea cycle for ridding the body of ammonia is the conversion of argininosuccinate to the amino acid arginine plus fumarate. Propose a mechanism for the reaction, and show the structure of arginine.
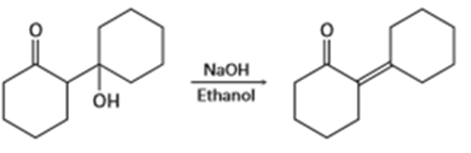
d) Tell whether each of the following reactions is likely to be SN1, SN2, E1, E1cB or E2:
What do you think about this solution?
We value your feedback to improve our textbook solutions.