Chapter 11: 44aE (page 350)
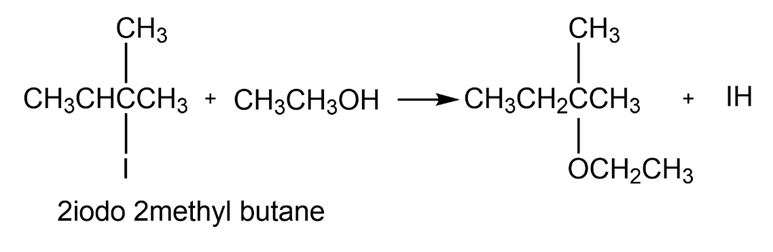
What effect would you expect the following changes to have on the rate of the reaction of ethanol with 2-iodo-2-methyl butane?
(a) The concentration of the halide is tripled.
Short Answer
Learning Materials
Features
Discover
Chapter 11: 44aE (page 350)
What effect would you expect the following changes to have on the rate of the reaction of ethanol with 2-iodo-2-methyl butane?
(a) The concentration of the halide is tripled.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Propose structures for compounds that fit the following descriptions:
(a) An alkyl halide that gives a mixture of three alkenes on E2 reaction
SN2 reactions take place with inversion of configuration, andSN1 reactions take place with racemization. The following substitution reaction, however, occurs with complete retention of configuration. Propose a mechanism.
.

Methyl esters () undergo a cleavage reaction to yield carboxylate ions plus iodomethane on heating with LiI in dimethylformamide:

The following evidence has been obtained: (1) The reaction occurs much faster in DMF than in ethanol. (2) The corresponding ethyl ester () cleaves approximately 10 times more slowly than the methyl ester. Propose a mechanism for the reaction. What other kinds of experimental evidence could you gather to support your hypothesis?
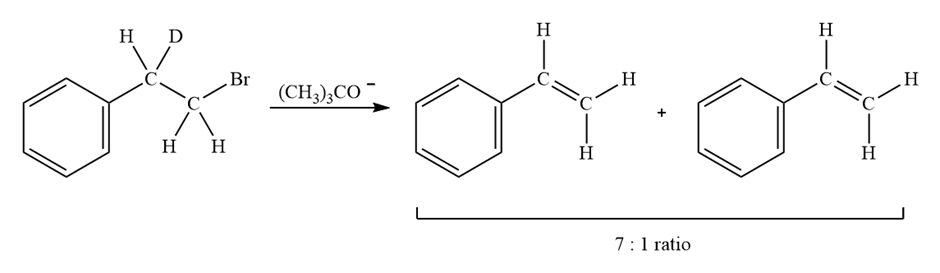
Treatment of 1-bromo-2-deuterio-2-phenylethane with strong base leads to a mixture of deuterated and nondeuterated phenylethylenes in an approximately 7;1 ratio. Explain
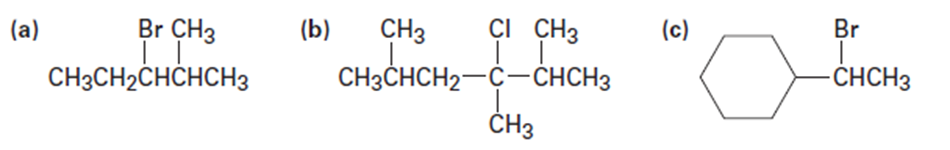
Question: Ignoring double-bond stereochemistry, what products would you expect from elimination reactions of the following alkyl halides? Which product will be the major product in each case?
What do you think about this solution?
We value your feedback to improve our textbook solutions.