Chapter 8: Q.8-8-66E-a (page 262)
How would you distinguish between the following pairs of compounds using simple chemical tests? Tell what you would do and what you would see.
(a) Cyclopentene and cyclopentane
Learning Materials
Features
Discover
Chapter 8: Q.8-8-66E-a (page 262)
How would you distinguish between the following pairs of compounds using simple chemical tests? Tell what you would do and what you would see.
(a) Cyclopentene and cyclopentane
All the tools & learning materials you need for study success - in one app.
Get started for free
Hydroxylation of cis-2-butene with yields a different product than hydroxylation of trans-2-butene. Draw the structure, show the stereochemistry of each product, and explain the difference between them.
One of the chain-termination steps that sometimes occurs to interrupt polymerization is the following reaction between two radicals. Propose a mechanism for the reaction, using fishhook arrows to indicate electron flow.
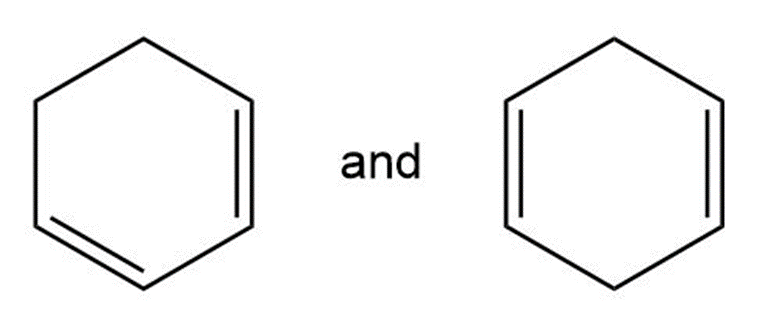
Using an oxidative cleavage reaction, explain how you would distinguish between the following two isomeric dienes:
An unknown hydrocarbon A with the formula reacts with 1 molar equivalent of over a palladium catalyst. Hydrocarbon A also reacts with to give diol B. When oxidized with in acidic solution, A gives two fragments. One fragment is propanoic acid, , and the other fragment is ketone C. What are the structures of A, B, and C? Write all reactions, and show your reasoning.
Question:Predict the products of the following reactions, showing both regiochemistry and stereochemistry where appropriate:
What do you think about this solution?
We value your feedback to improve our textbook solutions.