Chapter 2: Q.2-2-35_D (page 59)
Question: Calculate the formal charges on the atoms shown in red.
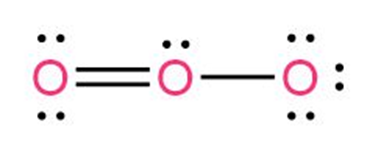
Short Answer
Answer
The formal charge on O at left is 0
The formal charge on O in the middle is +1
The formal charge on O at right is -1
Learning Materials
Features
Discover
Chapter 2: Q.2-2-35_D (page 59)
Question: Calculate the formal charges on the atoms shown in red.
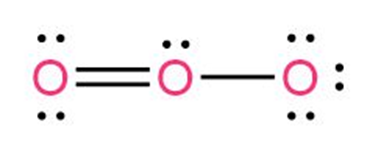
Answer
The formal charge on O at left is 0
The formal charge on O in the middle is +1
The formal charge on O at right is -1
All the tools & learning materials you need for study success - in one app.
Get started for free
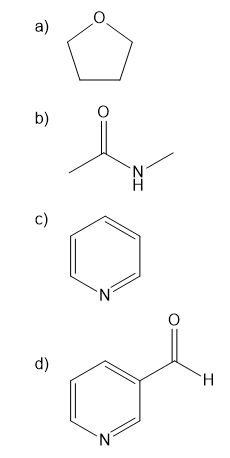
Question: Use curved arrows to draw the protonated form of each Lewis base below.
Question: Nitric acid (HNO3) reacts with ammonia (NH3) to yield ammonium nitrate. Write the reaction, and identify the acid, the base, the conjugate acid product, and the conjugate base product.
Draw the indicated number of resonance forms for each of the following
species:
(a)The methyl phosphate anion, CHOPO32-(3)
(b)The nitrate anion,NO3 (3)
(c)The allylcation,H2CPCHOCH2+(2)
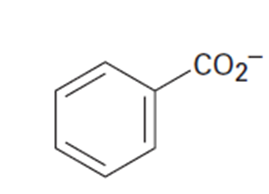
(d)The benzoate anion (4)
Thiamin diphosphate (TPP), a derivative of vitamin B1 required for glucose metabolism, is a weak acid that can be deprotonated by a base. Assign formal charges to the appropriate atoms in both TPP and its deprotonation product.

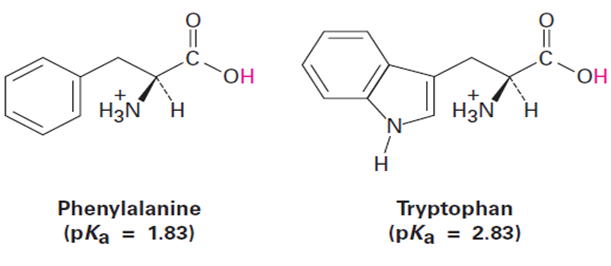
Question: The amino acid phenylalanine has pKa=1.83 , and tryptophan has pKa=1.83 Which is the stronger acid?
What do you think about this solution?
We value your feedback to improve our textbook solutions.