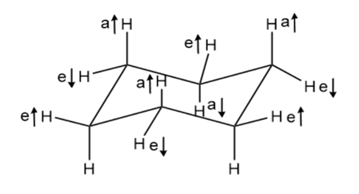
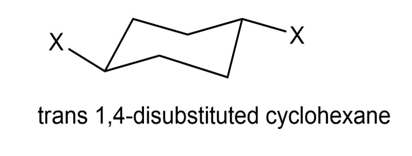
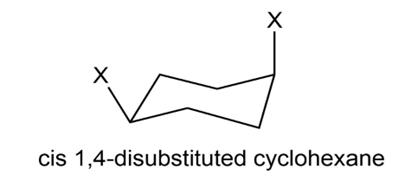
Chapter 4: Q4-38E (page 114)
Which is more stable, a 1, 4-trans disubstituted cyclohexane or its cis isomer?
Short Answer
1, 4-trans disubstituted cyclohexane is more stable than its cis isomer because 1, 4-cis disubstituted cyclohexane shows 1, 3 diaxial interaction.