Chapter 3: Q3-25E- b (page 88)
Predict the hybridization of the carbon atom in each of the following
Functional groups:
(b) Nitrile
Short Answer
(b) The hybridization of the carbon atom is sp hybridization.
Learning Materials
Features
Discover
Chapter 3: Q3-25E- b (page 88)
Predict the hybridization of the carbon atom in each of the following
Functional groups:
(b) Nitrile
(b) The hybridization of the carbon atom is sp hybridization.
All the tools & learning materials you need for study success - in one app.
Get started for free
For each of the following compounds, draw an isomer that has the same functional group.
a.
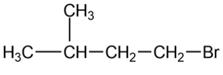
b.
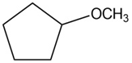
c.
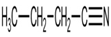
d.
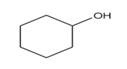
e.
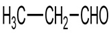
f.
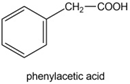
Draw a compound that:
Question: Make a graph of potential energy versus angle of bond rotation for propane,and assign values to the energy maxima.
Formaldehyde,, is known to all biologists because of its usefulness as a tissue preservative. When pure, formaldehyde trimerizes to give trioxane,, which, surprisingly enough, has no carbonyl groups. Only one monobromo derivative ()of trioxane is possible. Propose a structure for trioxane.
Sight along the C2-C1 bond of 2-methylpropane (isobutane).
(a) Draw a Newman projection of the most stable conformation.
What do you think about this solution?
We value your feedback to improve our textbook solutions.