Chapter 3: Q. 27 E-a (page 88)
Draw structures that meet the following descriptions (there are many
Possibilities):
(a) Three isomers with the formula
Learning Materials
Features
Discover
Chapter 3: Q. 27 E-a (page 88)
Draw structures that meet the following descriptions (there are many
Possibilities):
(a) Three isomers with the formula
All the tools & learning materials you need for study success - in one app.
Get started for free
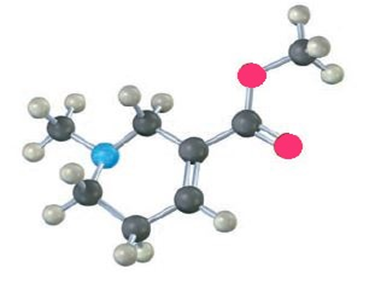
Identify the functional groups in the following model of arecoline, a veterinary drug used to control worms in animals. Convert the drawing into a line-bond structure and a molecular formula (red5O, blue5N).
Sight along the C2-C1 bond of 2-methylpropane (isobutane).
(a) Draw a Newman projection of the most stable conformation.
There are seven constitutional isomers with the formula . Drawas many as you can.
Propose structures that meet the following descriptions:
(c) Two isomeric disulfides with the formula C4H10S2
Question: The barrier to rotation about the C-C bond in bromoethane is 15 kJ/mol (3.6 kcal/mol).
(a) What energy value can you assign to an H←→Br eclipsing interaction?
(b) Construct a quantitative diagram of potential energy versus bond rotation for bromoethane.
What do you think about this solution?
We value your feedback to improve our textbook solutions.