Chapter 20: Q4P (page 653)
The for dichloroacetic acid is. Approximately what percentage of the acid is dissociated in a 0.10 M aqueous solution?
Short Answer
The percent dissociation of dichloroacetic acid in a 0.10 M aqueous solution is 43% .
Learning Materials
Features
Discover
Chapter 20: Q4P (page 653)
The for dichloroacetic acid is. Approximately what percentage of the acid is dissociated in a 0.10 M aqueous solution?
The percent dissociation of dichloroacetic acid in a 0.10 M aqueous solution is 43% .
All the tools & learning materials you need for study success - in one app.
Get started for free
Which would you expect to be a stronger acid, the lactic acid found in tired muscles or acetic acid? Explain.
Naturally occurring compounds called terpenoids, which we’ll discuss in section27-5, are biosynthesized by a pathway that involves loss of carbon dioxide from 3-phosphomevalonate 5-diphosphate to yield isopentenyl diphosphate. Use curved arrows to show the mechanism of this reaction.
Assume you have a mixture of naphthalene and benzoic acid that you want to separate. How might you take advantage of the acidity of one component in the mixture to effect a separation?
Rank the following compounds in order of increasing acidity. Don’t look at a table of data to help with your answer.
(a) Benzoic acid, p-methylbenzoic acid, p-chlorobenzoic acid
(b) p-Nitrobenzoic acid, acetic acid, benzoic acid
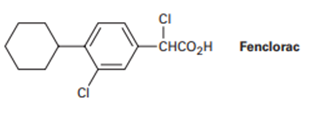
Propose a synthesis of the anti-inflammatory drug Fenclorac from phenyl cyclohexane.
What do you think about this solution?
We value your feedback to improve our textbook solutions.