Chapter 20: Q12P (page 667)
How might you carry out the following transformation? More than one step
is needed.
Short Answer
The below transformation is used to prepare 2-cyclopentyl-1-ethanol form
cyclopentylmethanol.
Learning Materials
Features
Discover
Chapter 20: Q12P (page 667)
How might you carry out the following transformation? More than one step
is needed.
The below transformation is used to prepare 2-cyclopentyl-1-ethanol form
cyclopentylmethanol.
All the tools & learning materials you need for study success - in one app.
Get started for free
Calculate the Ka's for the following acids:
(a) Citric acid, pKa = 3.14
(b) Tartaric acid, pKa = 2.98
How might you carry out the following transformation? More than one step is needed.

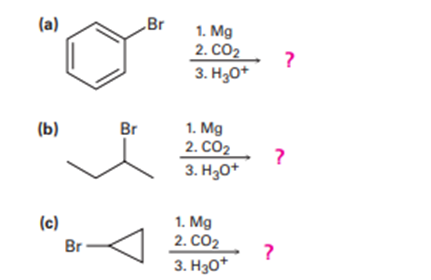
Predict the product and provide the mechanism for each reaction below.
The following pKavalues have been measured. Explain why a hydroxyl group in the para position decreases the acidity while a hydroxy group in the meta position increases the acidity.

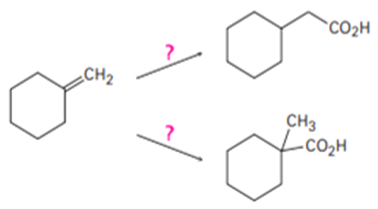
How would you carry out the following transformations?
What do you think about this solution?
We value your feedback to improve our textbook solutions.