Chapter 21: Q54 E (page 726)
The Following reactivity order has been found for the saponification of alkyl acetates by aqueous NaOH Explain.
Short Answer
The Order of saponification is
Learning Materials
Features
Discover
Chapter 21: Q54 E (page 726)
The Following reactivity order has been found for the saponification of alkyl acetates by aqueous NaOH Explain.
The Order of saponification is

All the tools & learning materials you need for study success - in one app.
Get started for free
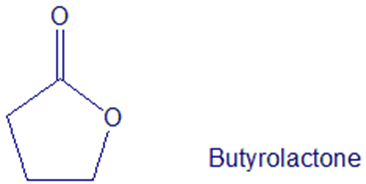
What product would you expect from the reaction of butyrolactone with LiAlH4? With DIBAH?
When ethyl benzoate is heated in methanol containing a small amount of HCL, Methyl benzoate is formed. Propose a mechanism for the reaction.
When an amide is formed from an acid chloride or an anhydride, two moles of base are required. However, when an ester is used as the starting material, only one equivalent of base is needed. Explain the reactivity in terms of basicity of the leaving groups.
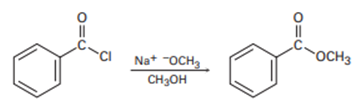
Show the mechanism of the following nucleophilic acyl substitution reaction, using curved arrows to indicate the electron flow in each step:
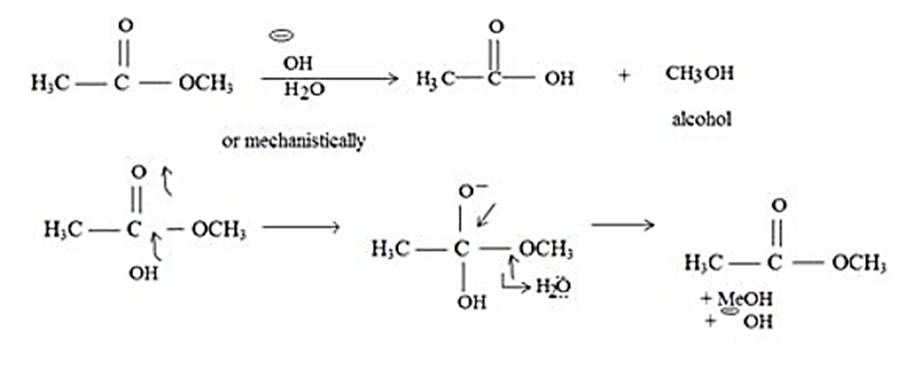
Why is the saponification of an ester irreversible? In other words, why doesn’t treatment of a carboxylic acid with an alkoxide ion yield an ester?
What do you think about this solution?
We value your feedback to improve our textbook solutions.