Chapter 21: Q21-23P (page 718)
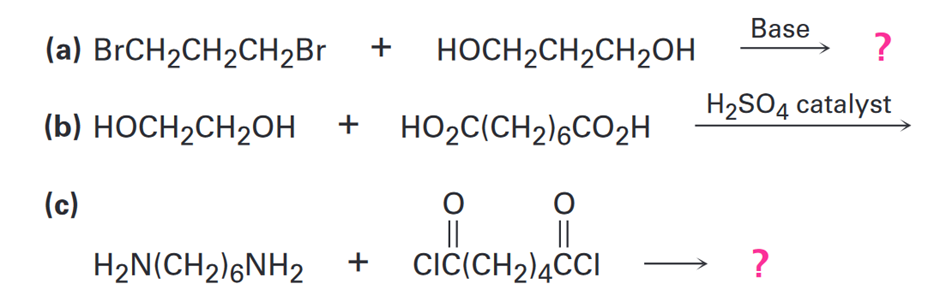
Draw structures of the step-growth polymers you would expect to obtain from the following reactions:
Short Answer
a.
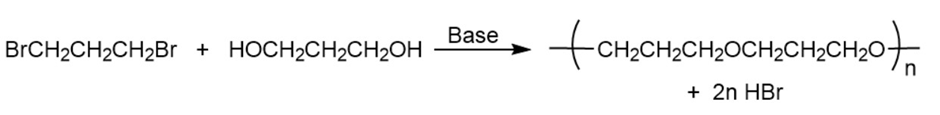
b.
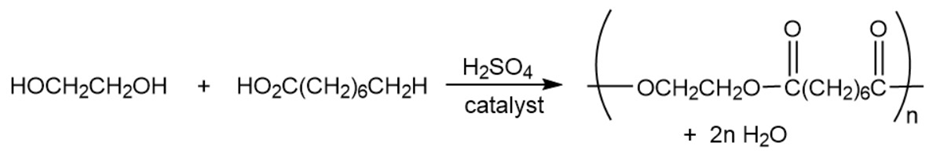
c.
Learning Materials
Features
Discover
Chapter 21: Q21-23P (page 718)
Draw structures of the step-growth polymers you would expect to obtain from the following reactions:

a.

b.

c.

All the tools & learning materials you need for study success - in one app.
Get started for free
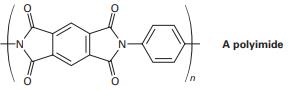
Question: 21-63 Polyimides with the structure shown are used as coatings on glass and plastics to improve scratch resistance. How would you synthesize a polyimide? (See Problem 21-39.)
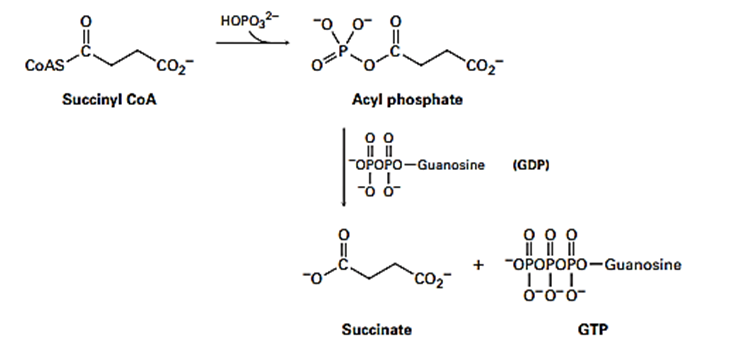
The hydrolysis of a biological thioester to the corresponding carboxylate is often more complex than the overall result might suggest. The conversion of succinyl CoA to succinate in the citric acid cycle, for instance, occurs by initial formation of an acyl phosphate, followed by reaction with guanosine diphosphate (GDP, a relative of adenosine diphosphate [ADP]) to give succinate and guanosine triphosphate (GTP, a relative of ATP). Suggest mechanisms for both steps.
21-43 The following reaction, called the benzilic acid rearrangement, takes place by typical carbonyl group reactions. Propose a mechanism (Ph=phenyl).
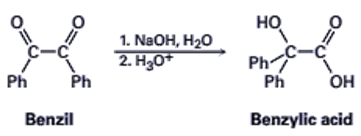
Propose structures for compounds that have the following formulas and IR absorptions:
(a) C6H12O2, 1735 cm-1
(b) C4H9NO, 1650 cm-1
(c) C4H5ClO, 1780 cm-1
How would you convert N-ethylbenzamide to each of the following products?
(a) Benzoic acid (b) Benzyl alcohol (c)
What do you think about this solution?
We value your feedback to improve our textbook solutions.