Chapter 21: Carboxylic Acid Derivatives: Nucleophilic Acyl Substitution Reactions
Q58 E
When ethyl benzoate is heated in methanol containing a small amount of HCL, Methyl benzoate is formed. Propose a mechanism for the reaction.
Q59 E
Butoxyycarbony azide, a reagent used in protein synthesis, is prepared by treating tert-butoxy carbonyl chloride with sodium azide. propose a pose mechanism for this reaction
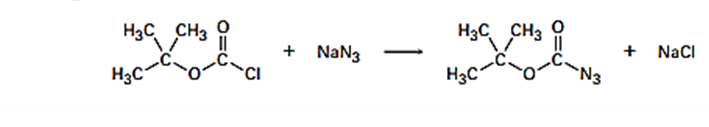
Q60 E
The step-growth polymer nylon 6 is prepared from caprolactam. The reaction involves the initial reaction of caprolactam with water to give an intermediate open-chain amino acid, followed by heating to form the polymer. Propose a mechanism for both steps, and show the structures of nylon 6.
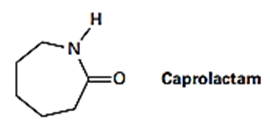
Q61 E
21-61 Qiana, a polyamide fiber with a silky texture, has the following structure. What are the monomer units used in the synthesis of Qiana?
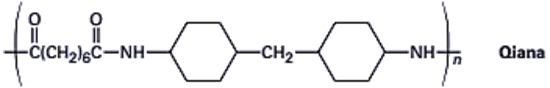
Q63E
Question: 21-63 Polyimides with the structure shown are used as coatings on glass and plastics to improve scratch resistance. How would you synthesize a polyimide? (See Problem 21-39.)
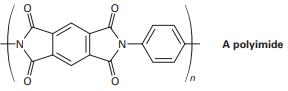
Q68E
21-68 When a carboxylic acid is dissolved in isotopically labeled water, thelabel rapidly becomes incorporated into both oxygen atoms of the carboxylic acid. Explain.
Q69E
21-69 We said in Section 21-6 that mechanistic studies on ester hydrolysishave been carried out using ethyl propanoate labeled with in theether-like oxygen. Assumethat labeled acetic acid is your onlysource of isotopic oxygen, and then propose a synthesis of the labeledethyl propanoate.
Q70E
21-70 Treatment of a carboxylic acid with trifluoroacetic anhydride leads to anunsymmetrical anhydride that rapidly reacts with alcohol to give an ester.
(a) Propose a mechanism for the formation of the unsymmetrical anhydride.
(b) Why is the unsymmetrical anhydride unusually reactive?
(c) Why does the unsymmetrical anhydride react as indicated ratherthan giving a trifluoroacetate ester plus carboxylic acid?
Q71E
21-71 Butacetin is an analgesic (pain-killing) agent that is synthesized commercially from p-fluoronitrobenzene. Propose a synthesis.
Q76P
Draw the structure of the polymer you would expect to obtain from
reaction of dimethyl terephthalate with a triol such as glycerol. Whatstructural feature would this new polymer have that was not present inDacron (Table 21-2)? How do you think this new feature might affectthe properties of the polymer?

