Chapter 23: Q37E (page 783)
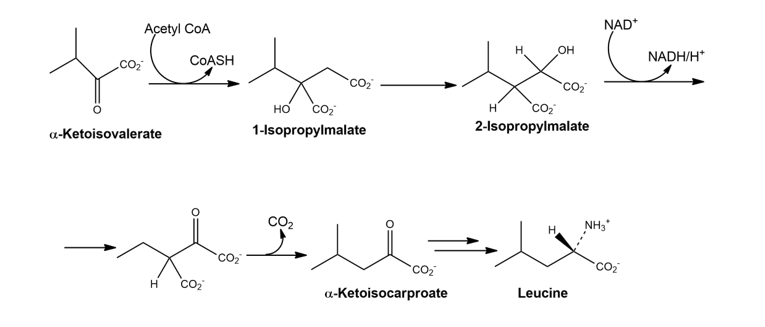
The amino acid leucine is biosynthesized from α-ketoisovalerate by the following sequence of steps. Show the mechanism of each.
Short Answer
The synthesis of leucine takes place from α-ketoisovalerate.
Learning Materials
Features
Discover
Chapter 23: Q37E (page 783)
The amino acid leucine is biosynthesized from α-ketoisovalerate by the following sequence of steps. Show the mechanism of each.

The synthesis of leucine takes place from α-ketoisovalerate.
All the tools & learning materials you need for study success - in one app.
Get started for free
Predict the product(s) and provide the mechanism for each reaction below.

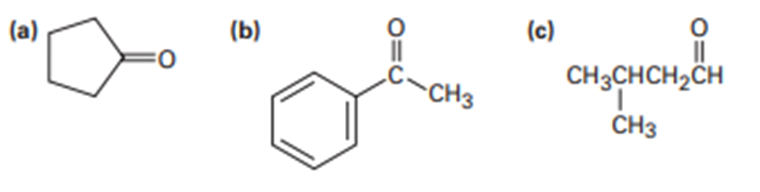
What enone product would you expect from aldol condensation of each of the following compounds?
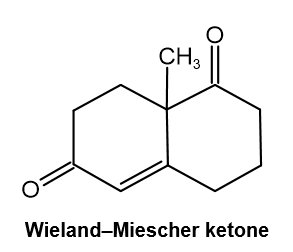
The so-called Wieland–Miescher ketone is a valuable starting material used in the synthesis of steroid hormones. How might you prepare it from 1,3-cyclohexanedione?
Which of the following compounds would you expect to undergo aldol self-condensation? Show the product of each successful reaction.
(a) Trimethyl acetaldehyde
(b) Cyclobutanone
(c) Benzophenone (diphenyl ketone)
(d) 3-Pentanone
(e) Decanal
(f) 3-Phenyl-2-propenal
Predict the addition product for each reaction below and provide the mechanism.

What do you think about this solution?
We value your feedback to improve our textbook solutions.