Chapter 23: Q23-7P (page 761)
Show how you would synthesize the following compound using an aldol reaction:
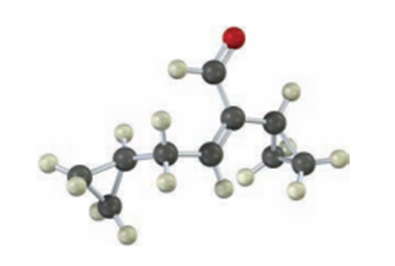
Short Answer
The name of the given compound is:Cyclopropyl Acetaldehyde.
Learning Materials
Features
Discover
Chapter 23: Q23-7P (page 761)
Show how you would synthesize the following compound using an aldol reaction:

The name of the given compound is:Cyclopropyl Acetaldehyde.
All the tools & learning materials you need for study success - in one app.
Get started for free
Dieckmann cyclization of diethyl 3-methylheptanedioate gives a mixture of two𝛃-keto ester products. What are their structures, and why is a mixture formed?
Which of the following compounds would you expect to undergo aldol self-condensation? Show the product of each successful reaction.
(a) Trimethyl acetaldehyde
(b) Cyclobutanone
(c) Benzophenone (diphenyl ketone)
(d) 3-Pentanone
(e) Decanal
(f) 3-Phenyl-2-propenal
The Mannich reaction of a ketone, an amine, and an aldehyde is one of the few three-component reactions in organic chemistry. Cyclohexanone, for example, reacts with dimethylamine and acetaldehyde to yield an amino ketone. The reaction takes place in two steps, both of which are typical carbonyl-group reactions.
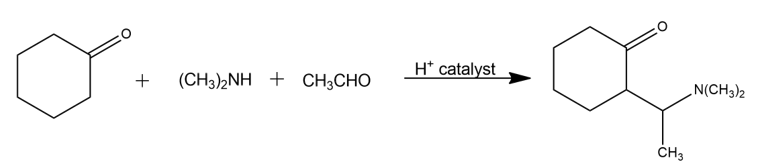
(a)The first step is reaction between the aldehyde and the amine to yield an intermediate iminium ion plus water. Propose a mechanism, and show the structure of the intermediate iminium ion.
(b)The second step is reaction between the iminium ion intermediate and the ketone to yield the final product. Propose a mechanism.
What product would you obtain from a base-catalyzed Michael reaction of 3-buten-2-one with each of the following nucleophilic donors?
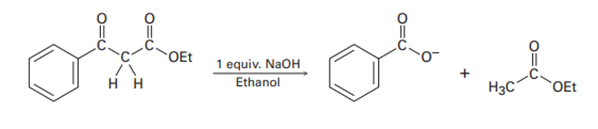
As shown in Figures 23-4, the Claisen reaction is reversible. That is, a𝛃-keto ester can be cleaved by base into two fragments. Using curved arrows to indicate electron flow, show the mechanism by which this cleavage occurs.
What do you think about this solution?
We value your feedback to improve our textbook solutions.