Chapter 23: Q10P (page 764)
What product would you expect to obtain from the base treatment of 1,6-cyclo-decanedione?
Short Answer
It gives seven-member ring fused with the five-member ring.
Learning Materials
Features
Discover
Chapter 23: Q10P (page 764)
What product would you expect to obtain from the base treatment of 1,6-cyclo-decanedione?
It gives seven-member ring fused with the five-member ring.
All the tools & learning materials you need for study success - in one app.
Get started for free
What product would you expect from the following mixed Claisen-like reaction?
Intramolecular aldol cyclization of 2,5-heptanedione with aqueous NaOH yields a mixture of two enone products in the approximate ratio 9;1. Write their structures, and show how each is formed.
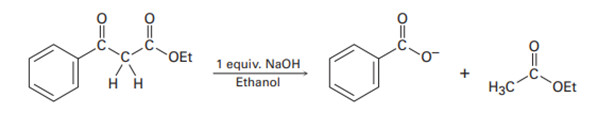
As shown in Figures 23-4, the Claisen reaction is reversible. That is, a𝛃-keto ester can be cleaved by base into two fragments. Using curved arrows to indicate electron flow, show the mechanism by which this cleavage occurs.
What enone product would you expect from aldol condensation of each of the following compounds?

Show the products you would expect to obtain by Claisen condensation of the following esters:(a) (b) Ethyl phenylacetate (c) Ethyl cyclohexyl acetate
What do you think about this solution?
We value your feedback to improve our textbook solutions.