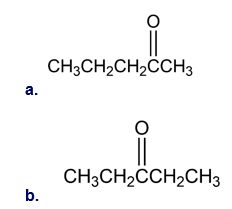
Chapter 9: Q9-7P b (page 272)
How would you prepare the following carbonyl compounds starting from an alkyne (reddish-brown Br)?
b.
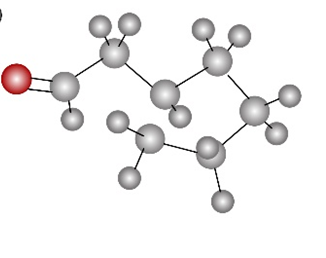
Short Answer
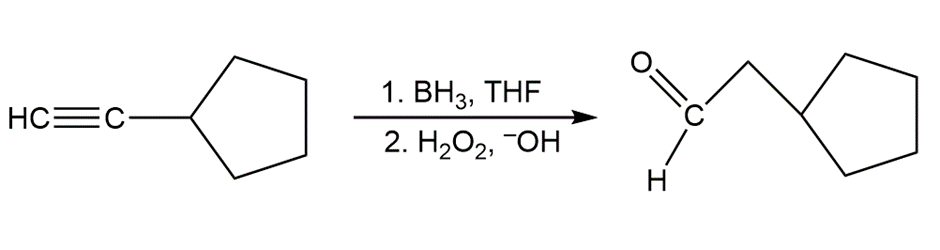
Learning Materials
Features
Discover
Chapter 9: Q9-7P b (page 272)
How would you prepare the following carbonyl compounds starting from an alkyne (reddish-brown Br)?
b.


All the tools & learning materials you need for study success - in one app.
Get started for free
What products would you obtain by hydration of the following alkynes?
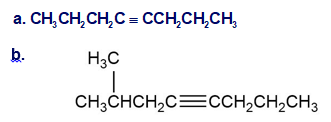
Synthesize the following compounds using 1-butyne as the only source of carbon along with any inorganic reagents you need. More than one step may be needed.(a) 1,1,2,2- Tetrachlorobutane(b) 1,1- Dichloro-2-ethylcyclopropane
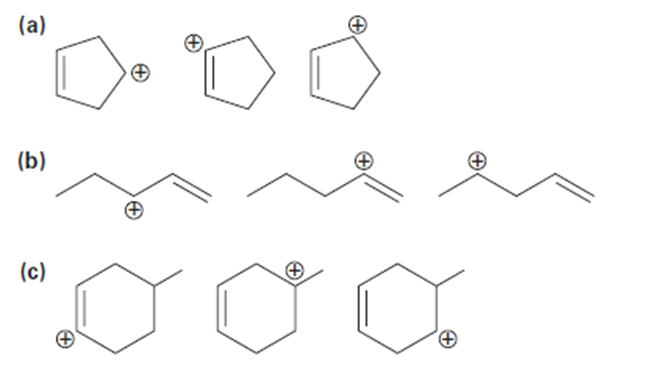
Arrange the carbocations below in order of increasing stability.
What alkynes would you start with to prepare the following ketones?
The sex attractant given off by the common housefly is an alkene named muscalure. Propose a synthesis of muscalure starting from acetylene and any alkyl halides needed. What is the IUPAC name for muscalure?
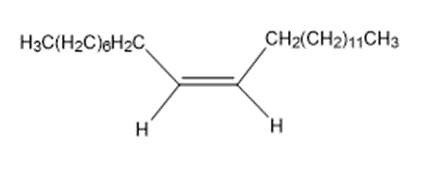
Muscalure
What do you think about this solution?
We value your feedback to improve our textbook solutions.