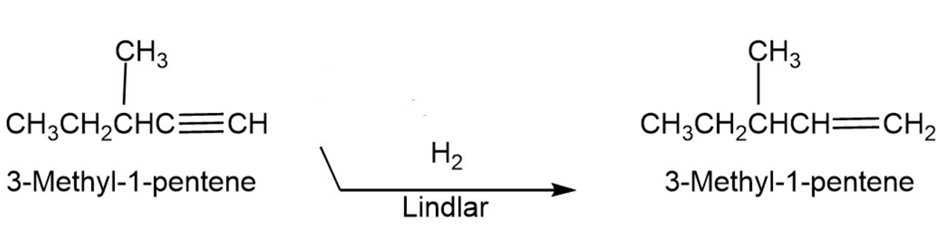
Chapter 9: Q8 P-c (page 274)
Question:Using any alkyne needed, how would you prepare the following alkenes?
(c) 3-Methyl-1-pentene
Short Answer
Learning Materials
Features
Discover
Chapter 9: Q8 P-c (page 274)
Question:Using any alkyne needed, how would you prepare the following alkenes?
(c) 3-Methyl-1-pentene

All the tools & learning materials you need for study success - in one app.
Get started for free
A cumulene is a compound with three adjacent double bonds. Draw an orbital picture of a cumulene. What kind of hybridization do the two central carbon atoms have? What is the geometric relationship of the substituents on one end to the substituents on the other end? What kind of isomerism is possible? Make a model to help see the answer.
A cumulene
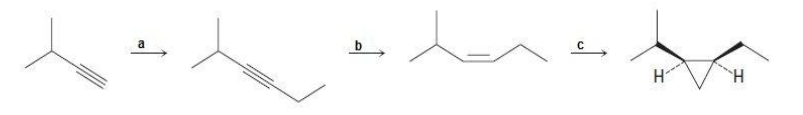
Question: Identify the reagents a–c in the following scheme:
Using any alkyne needed, how would you prepare the following alkenes?
(a) trans-2-Octene
Using any alkyne needed, how would you prepare the following alkenes?
(b) cis-3-Heptene
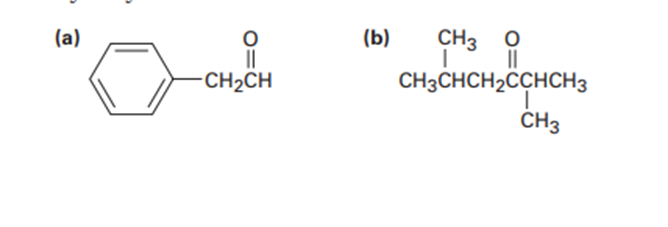
What alkyne would you start with to prepare each of the following compounds by a hydroboration–oxidation reaction?
What do you think about this solution?
We value your feedback to improve our textbook solutions.