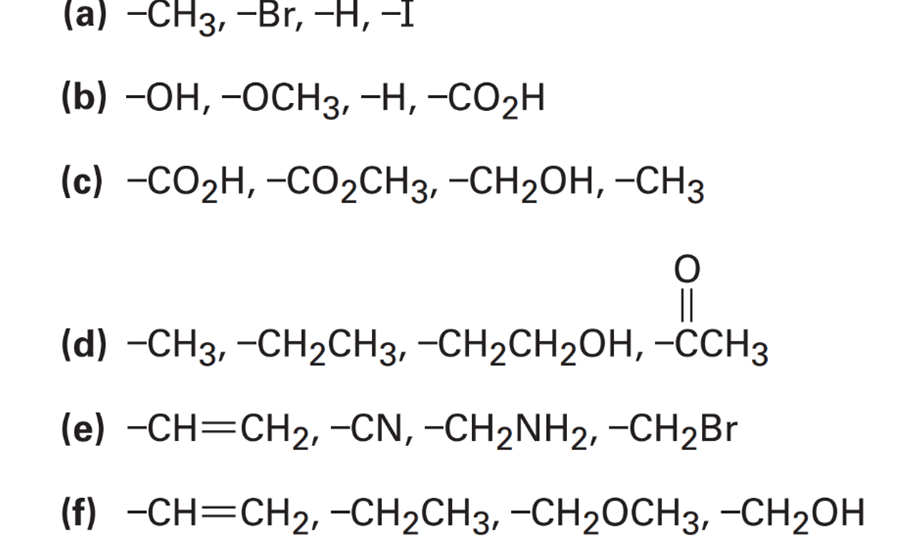
Chapter 7: Q36E (page 219)
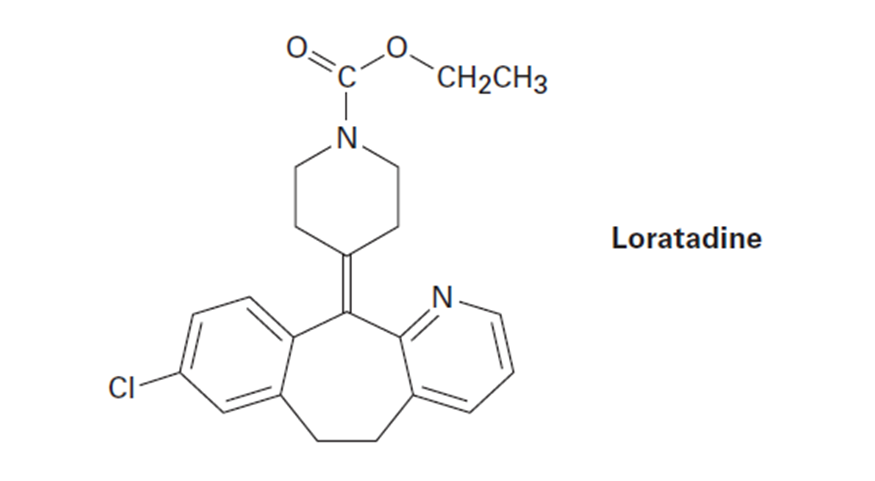
Question: Loratadine, marketed as an antiallergy medication under the name Claritin, has four rings, eight double bonds, and the formula C22H?CIN2O2 . How many hydrogens does loratadine have? (Calculate your answer; don’t count hydrogens in the structure.)
Short Answer
Answer
The number of hydrogens in loratidine is 23.