Chapter 17: Q50P (page 567)
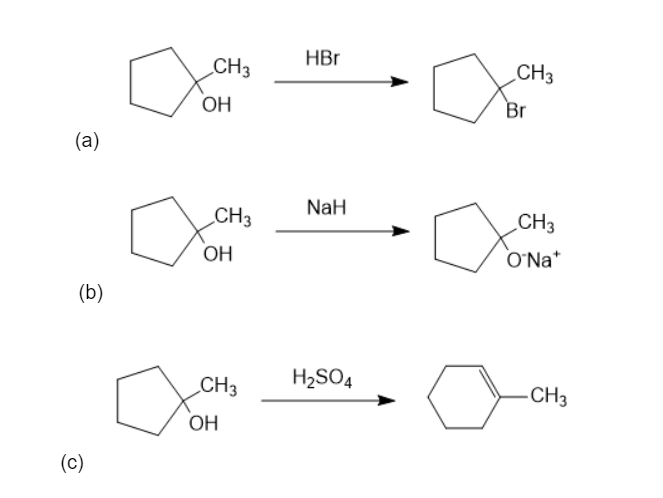
What products would you expect to obtain from reaction of 1-methylcyclohexanol with the following reagents?
(a) HBr (b) NaH (c) H2SO4 (d) Na2Cr2O7
Short Answer
Learning Materials
Features
Discover
Chapter 17: Q50P (page 567)
What products would you expect to obtain from reaction of 1-methylcyclohexanol with the following reagents?
(a) HBr (b) NaH (c) H2SO4 (d) Na2Cr2O7


All the tools & learning materials you need for study success - in one app.
Get started for free
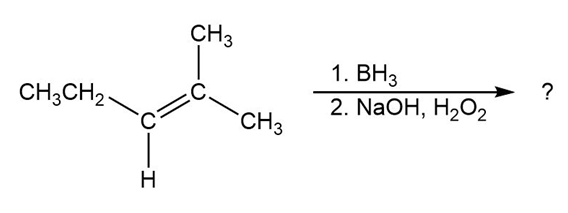
Question: Predict the products of the following reactions:
(a)
(b)
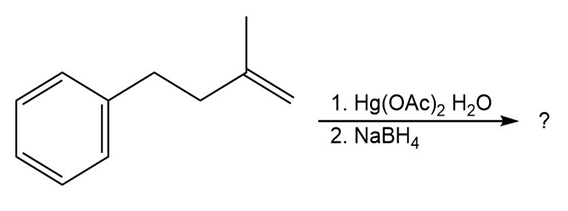
(c)
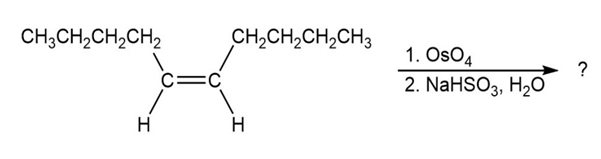
Question:The trimethylsilyl (TMS) protecting group is one of several silicon protecting groups for alcohols. For each reaction, draw the mechanism for the protection of (R)-3-bromo-1-butanol with the following silyl chlorides, using triethylamine as the base:
(a) tert-butyldimethylsilyl chloride (TBS-Cl)
(b) triisopropylsilyl chloride (TIPS-Cl)
(c) triethylsilyl chloride (TES-Cl)
The trimethylsilyl (TMS) protecting group is one of several silicon protecting groups for alcohols. For each reaction, draw the mechanism for the protection of (R)-3-bromo-1-butanol with the following silyl chlorides, using triethylamine as the base:
(a) tert-butyldimethylsilyl chloride (TBS-Cl)
(b) triisopropylsilyl chloride (TIPS-Cl)
(c) triethylsilyl chloride (TES-Cl)
Question:Acid-catalyzed dehydration of 2,2-dimethylcyclohexanol yields a mixture of 1,2-methylcyclohexane and isopropylidenecyclopentane. Propose a mechanism to account for the formation of both products.

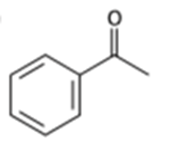
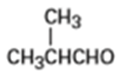
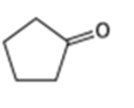
What alcohols would give the following products on oxidation?
(a)
(b)
(c)
What do you think about this solution?
We value your feedback to improve our textbook solutions.