Chapter 6: Q54P (page 280)
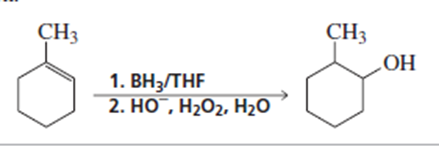
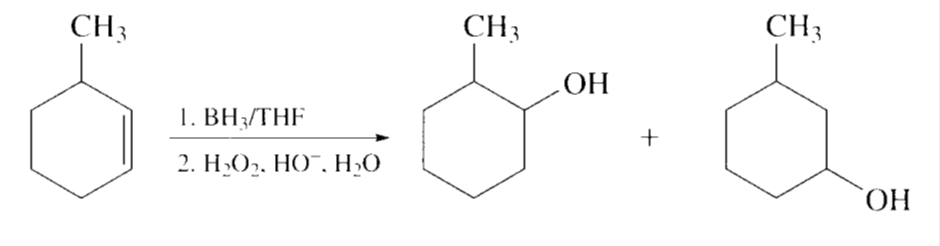
Explain why 3-methylcyclohexene should notbe used as the starting material in Problem 52b.
Short Answer
Two products will form and we get less desired product.
Learning Materials
Features
Discover
Chapter 6: Q54P (page 280)
Explain why 3-methylcyclohexene should notbe used as the starting material in Problem 52b.

Two products will form and we get less desired product.

All the tools & learning materials you need for study success - in one app.
Get started for free
What will be the product of the preceding reaction if HBr is used in place of Br2?
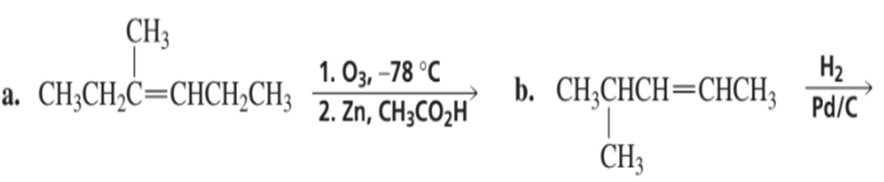
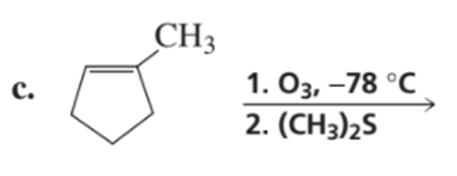
a.What alkene is required to synthesize each of the following compounds?
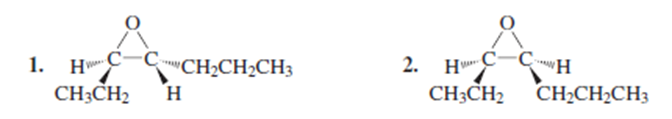
b. What other epoxide would be formed?
c. Assign an Ror Sconfiguration to each asymmetric center.
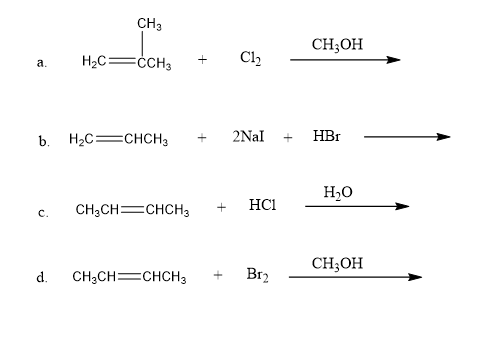
Each of the following reactions has two nucleophiles that could add to the intermediate formed by the reaction of the alkene with an electrophile. What is the major product of each reaction
Which compound is hydrated more rapidly?

What are the products of the following reactions? Indicate whether each reaction is an oxidation or a reduction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.