Chapter 6: Q97P (page 287)
Which compound is hydrated more rapidly?

Short Answer
The 1st compound is hydrated faster due to the stability of carbocation in transition state.
Learning Materials
Features
Discover
Chapter 6: Q97P (page 287)
Which compound is hydrated more rapidly?

The 1st compound is hydrated faster due to the stability of carbocation in transition state.
All the tools & learning materials you need for study success - in one app.
Get started for free
Propose a mechanism for the following reaction:

The reaction of 2-ethyl-1-pentene with Br2, with H2 + Pd/C, or with R2BH/THF followed by aqueous HO- + H2O2 leads to a racemic mixture. Explain why a racemic mixture is obtained in each case.
What is the major product of each of the following reactions?
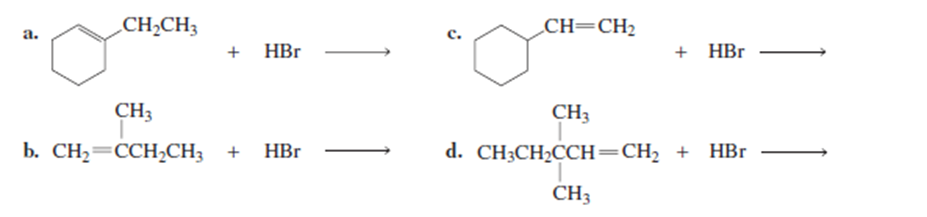
a. How many s bond orbitals are available for overlap with the vacant p orbital in the methyl cation?
b. Which is more stable: a methyl cation or an ethyl cation? Why?
a. What is the product of the reaction of fumarate and H2O when H2SO4 is used as a catalyst instead of fumarase?
b. What is the product of the reaction of maleate and H2O when H2SO4 is used as a catalyst instead of fumarase?
What do you think about this solution?
We value your feedback to improve our textbook solutions.