Chapter 9: Q68P (page 451)
Which member of each pair is a better nucleophile in methanol?
a. H2O or HO-
b. NH3 or H2O
c. H2O or H2S
d. HO- or HS-
e. I- or Br-
f. Cl-or Br-
Short Answer
- HO-
- NH3
- H2S
- HS-
- I-
- Br-
Learning Materials
Features
Discover
Chapter 9: Q68P (page 451)
Which member of each pair is a better nucleophile in methanol?
a. H2O or HO-
b. NH3 or H2O
c. H2O or H2S
d. HO- or HS-
e. I- or Br-
f. Cl-or Br-
All the tools & learning materials you need for study success - in one app.
Get started for free
Why do cis-1-bromo-2-ethylcyclohexane and trans-1-bromo-2-ethylcyclohexane form different major products when they undergo an E2 reaction?
a. Which reacts faster in an E2 reaction: 3-bromocyclohexene or Bromo cyclohexane?
b. Which reacts faster in an E1 reaction?
What percentage of the reaction described in Problem 31 takes place by the E2 pathway when [HO-] = 0.0025 M?
Draw the structure of DDE.
What is the major elimination product obtained from the reaction of each of the following alkyl halides with hydroxide ions?
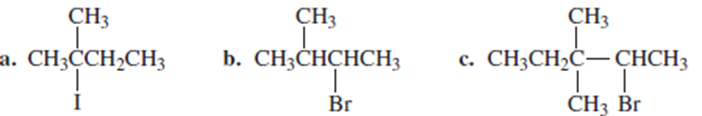
What do you think about this solution?
We value your feedback to improve our textbook solutions.