Chapter 9: Q66P (page 448)
After a proton is removed from the OH group, which compound in each pair forms a cyclic ether more rapidly?
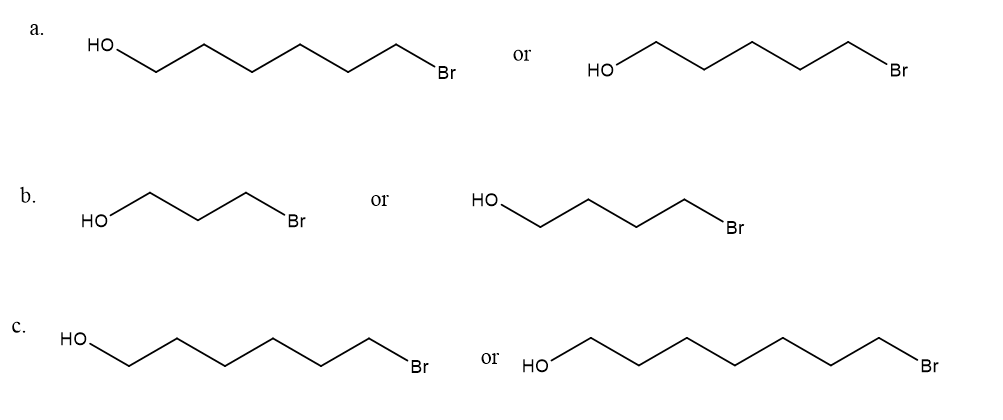
Short Answer
These compounds form a cyclic ether-
Learning Materials
Features
Discover
Chapter 9: Q66P (page 448)
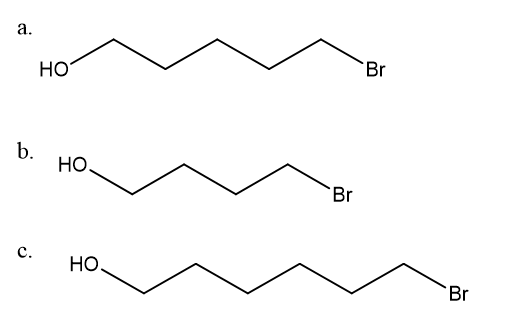
After a proton is removed from the OH group, which compound in each pair forms a cyclic ether more rapidly?

These compounds form a cyclic ether-

All the tools & learning materials you need for study success - in one app.
Get started for free
Explain how the following changes affect the rate of the reaction of 2-bromo-2-methylbutane with methanol:
a. The alkyl halide is changed to 2-chloro-2-methylbutane.
b. The alkyl halide is changed to 2-chloro-3-methylbutane.
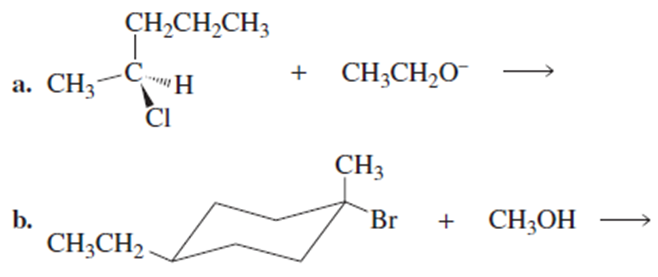
Draw the configuration(s) of the substitution product(s) formed from the reaction of the following compounds with the indicated nucleophiles:
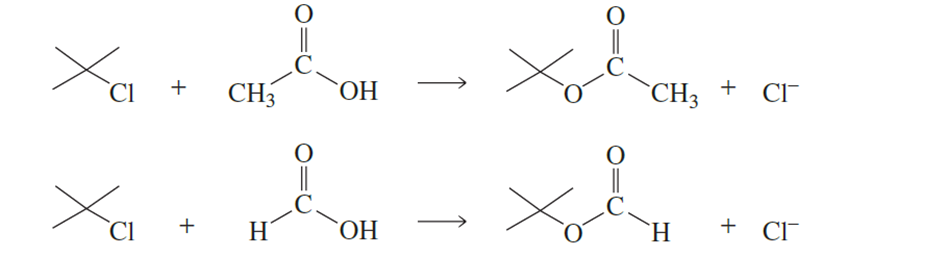
tert-Butyl chloride undergoes solvolysis in both acetic acid and formic acid. Solvolysis occurs 5000 times faster in one of these two solvents than in
the other. In which solvent is solvolysis faster? Explain your answer. (Hint: Formic acid is more polar than acetic acid.)
A chemist wanted to synthesize the aesthetic 2-ethoxy-2-methylpropane. He used ethoxide ion and 2-chloro-2-methylpropane for his synthesis and ended up with no ether. What was the product of his synthesis? What reagents should he have used?
Indicate whether each of the following solvents is protic or aprotic:
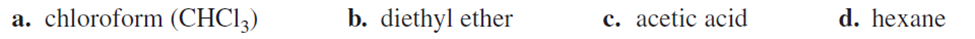
What do you think about this solution?
We value your feedback to improve our textbook solutions.