Chapter 9: Q20P (page 411)
Draw the configuration(s) of the substitution product(s) formed from the reaction of the following compounds with the indicated nucleophiles:
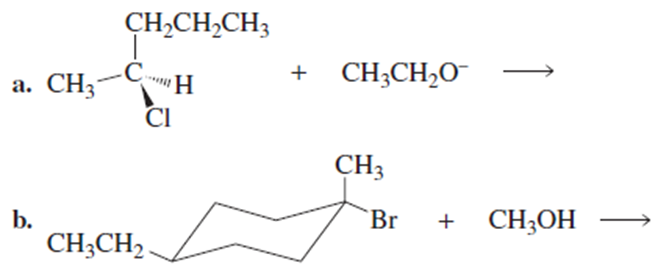
Short Answer
a)
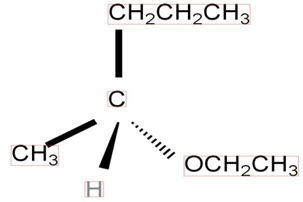
It follows SN2 mechanism.
b.
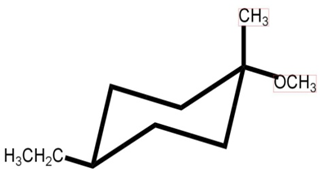
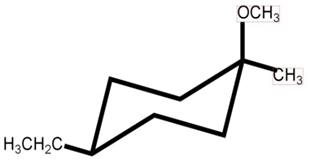
It follows SN2 mechanism.
Learning Materials
Features
Discover
Chapter 9: Q20P (page 411)
Draw the configuration(s) of the substitution product(s) formed from the reaction of the following compounds with the indicated nucleophiles:

a)

It follows SN2 mechanism.
b.


It follows SN2 mechanism.
All the tools & learning materials you need for study success - in one app.
Get started for free
A chemist wanted to synthesize the aesthetic 2-ethoxy-2-methylpropane. He used ethoxide ion and 2-chloro-2-methylpropane for his synthesis and ended up with no ether. What was the product of his synthesis? What reagents should he have used?
a. Which is a stronger base: RO- or RS-?
b. Which is a better nucleophile in an aqueous solution?
c. Which is a better nucleophile in DMSO?
Two elimination products are obtained from the following E2 reaction
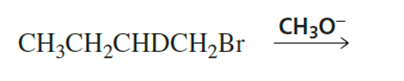
a. What are the elimination products?
b. Which is formed in greater yield? (Hint:See Problem 91.)
Draw the products of each of the following SN2/E2 reactions. If the products can exist as stereoisomers, show which stereoisomers are formed.
a.(3S,4S)-3-bromo-4-methylhexane + CH3O-
b.(3R,4R)-3-bromo-4-methylhexane + CH3O-
c.(3S,4R)-3-bromo-4-methylhexane + CH3O-
d.(3R,4S)-3-bromo-4-methylhexane + CH3O-
Which member in each pair in Problem 68 is a better leaving group?
What do you think about this solution?
We value your feedback to improve our textbook solutions.