Chapter 9: Q32P (page 422)
What percentage of the reaction described in Problem 31 takes place by the E2 pathway when [HO-] = 0.0025 M?
Short Answer
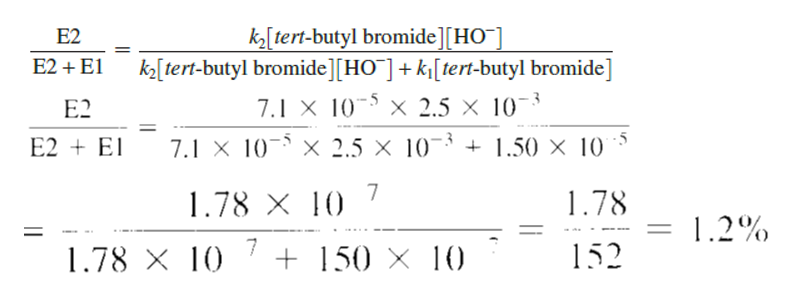
Learning Materials
Features
Discover
Chapter 9: Q32P (page 422)
What percentage of the reaction described in Problem 31 takes place by the E2 pathway when [HO-] = 0.0025 M?
All the tools & learning materials you need for study success - in one app.
Get started for free
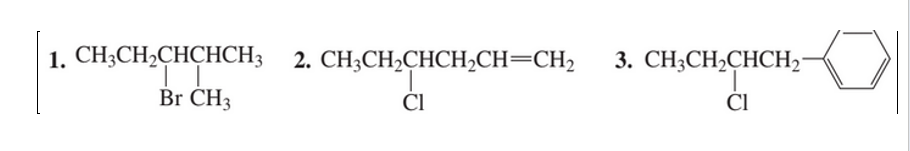
a. What is the major product obtained when each of the following compounds undergoes an E2 reaction with methoxide ion ? Show the configuration of the product.
b. Does the product obtained depend on whether you start with the R or S enantiomer of the reactant?
Question:Draw the elimination products for each of the following E2 reactions; if the products can exist as stereoisomers, indicate which stereoisomers are obtained.
a. (2S,3S)-2-chloro-3-methylpentane + high concentration of CH3O-
b.(2S,3R)-2-chloro-3-methylpentane + high concentration of CH3O-
c.(2R,3S)-2-chloro-3-methylpentane + high concentration of CH3O-
d.(2R,3R)-2-chloro-3-methylpentane + high concentration of CH3O-
e.3-chloro-3-ethyl-2,2-dimethylpentane + high concentration of CH3CH2O-
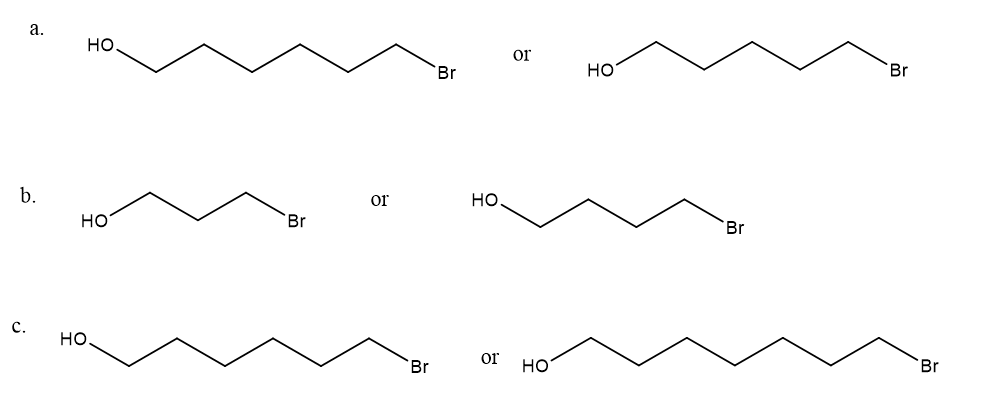
After a proton is removed from the OH group, which compound in each pair forms a cyclic ether more rapidly?
a. Identify the substitution products that form when 2-bromo-2-methylpropane is dissolved in a mixture of 80% ethanol and 20% water.
b. Explain why the same products are obtained when 2-chloro-2-methylpropane is dissolved in a mixture of 80% ethanol and 20% water.
The reaction of chloromethane with hydroxide ion at 30°C has a ΔG° value of -21.7 kcal/mol. What is the equilibrium constant for the reaction?
What do you think about this solution?
We value your feedback to improve our textbook solutions.