Chapter 9: Q46P (page 435)
What products will be obtained from the E1 reaction of the alkyl halides in Problem 45?
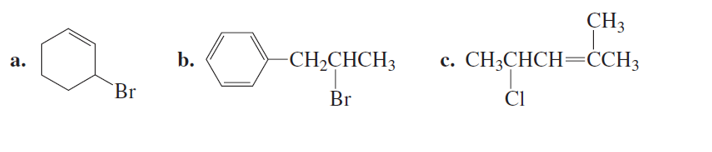
Short Answer
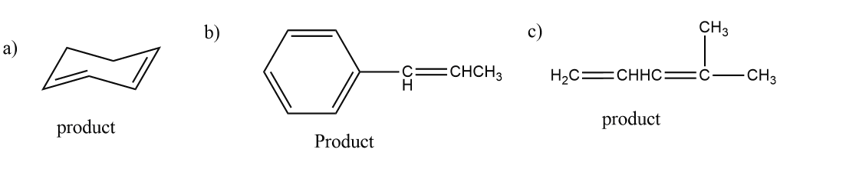
The formed products are,
Learning Materials
Features
Discover
Chapter 9: Q46P (page 435)
What products will be obtained from the E1 reaction of the alkyl halides in Problem 45?
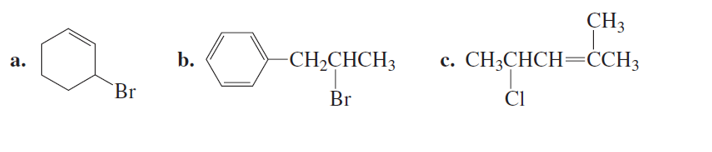
The formed products are,

All the tools & learning materials you need for study success - in one app.
Get started for free
a. Identify the substitution products that form when 2-bromo-2-methylpropane is dissolved in a mixture of 80% ethanol and 20% water.
b. Explain why the same products are obtained when 2-chloro-2-methylpropane is dissolved in a mixture of 80% ethanol and 20% water.
How will the rate of the reaction between bromomethane and hydroxide ion be affected if the following changes in concentration are made?
a. The concentration of the alkyl halide is not changed and the concentration of the nucleophile is tripled.
b. The concentration of the alkyl halide is cut in half and the concentration of the nucleophile is not changed.
c. The concentration of the alkyl halide is cut in half and the concentration of the nucleophile is doubled.
Draw the substitution products for each of the following reactions; if the products can exist as stereoisomers, show what stereoisomers are obtained:
a. ( R )-2-bromopentane + CH3O-
b. ( R )-3-bromo-3-methylheptane + CH3OH
c. benzyl chloride + CH3CH2OH
d. allyl chloride + CH3OH
e. 1-bromo-2-butene + CH3O-
f. 1-bromo-2-butene + CH3OH
Explain how the following changes affect the rate of the reaction of 2-bromo-2-methylbutane with methanol:
a. The alkyl halide is changed to 2-chloro-2-methylbutane.
b. The alkyl halide is changed to 2-chloro-3-methylbutane.
Draw the major product obtained when each of the following alkyl halides undergoes an E2 reaction:

What do you think about this solution?
We value your feedback to improve our textbook solutions.