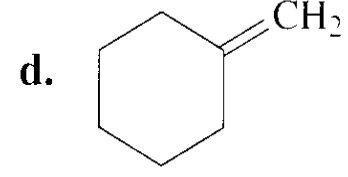
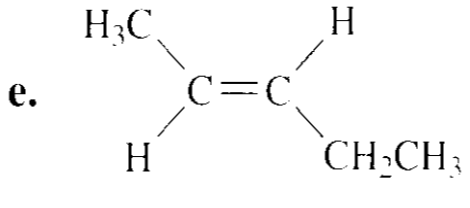
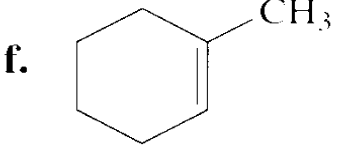
Chapter 9: Q83P (page 452)
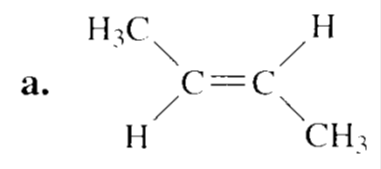
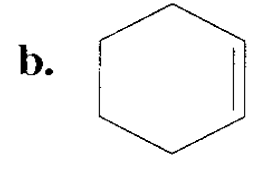
Draw the major product obtained when each of the following alkyl halides undergoes an E2 reaction:
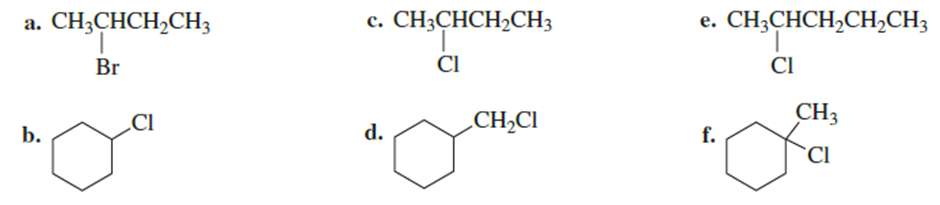
Short Answer
Learning Materials
Features
Discover
Chapter 9: Q83P (page 452)
Draw the major product obtained when each of the following alkyl halides undergoes an E2 reaction:







All the tools & learning materials you need for study success - in one app.
Get started for free
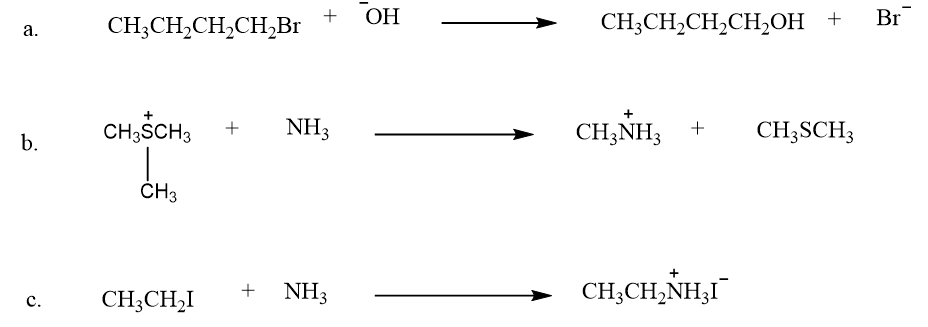
How will the rate of each of the following SN2reactions change if it is carried out in a more polar solvent?
Rank the following alkyl bromides from most reactive to least reactive in an SN2 reaction:
1-bromo-2-methylbutane, 1-bromo-3-methylbutane, 2-bromo-2-methylbutane, and 1-bromopentane.
cis-4-Bromocyclohexanol and trans-4-bromocyclohexanol form the same elimination productbut a different substitution product when they reactwith HO-.
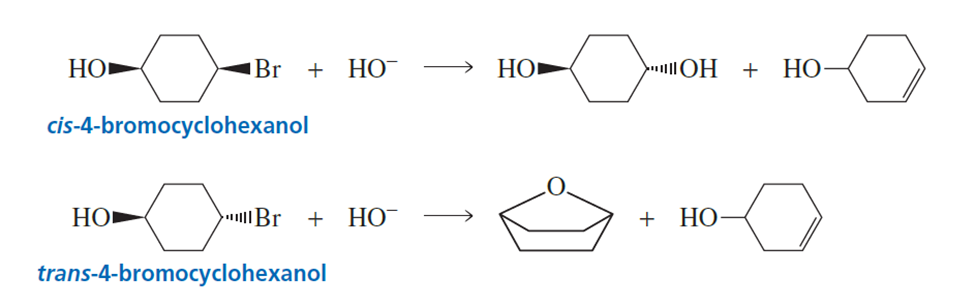
a. Why do they form the same elimination product?
b. Explain, by showing the mechanisms, why different substitution products are obtained.
c. How many stereoisomers does each of the elimination and substitution reactions form?
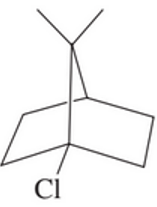
Explain why the following alkyl halide does not undergo a substitution reaction, regardless of the base that is used.
Rank the following alkyl halides from most reactive to least reactive in an SN1 reaction:
2-bromo-2-methylpentane, 2-chloro-2-methylpentane, 3-chloropentane, and 2-iodo-2-methylpentane.
What do you think about this solution?
We value your feedback to improve our textbook solutions.