Chapter 9: Q47P (page 435)
Draw the elimination products that are formed when 3-bromo-3-methyl-1-butene reacts with
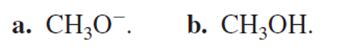
Short Answer
The products are given below,
Learning Materials
Features
Discover
Chapter 9: Q47P (page 435)
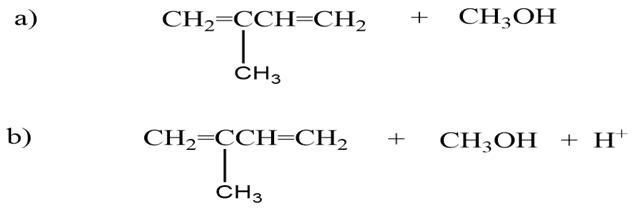
Draw the elimination products that are formed when 3-bromo-3-methyl-1-butene reacts with
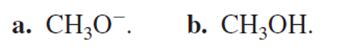
The products are given below,

All the tools & learning materials you need for study success - in one app.
Get started for free
Which compound is more reactive in an SN1 reaction? In each case, you can assume that both alkyl halides have the same stability.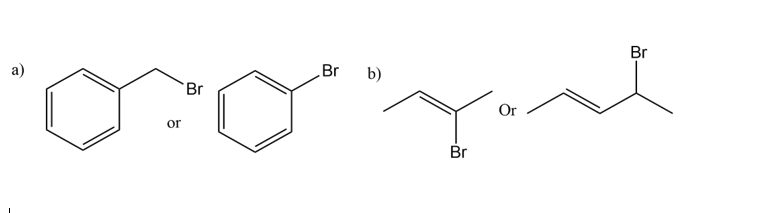
What products are formed from the following reactions?

Draw the stereoisomers that are formed from the following SN1 reactions:
a. 3-bromo-3-methylpentane and methanol
b. 3-chloro-3-methylhexane and methanol
tert-Butyl chloride undergoes solvolysis in both acetic acid and formic acid. Solvolysis occurs 5000 times faster in one of these two solvents than in
the other. In which solvent is solvolysis faster? Explain your answer. (Hint: Formic acid is more polar than acetic acid.)
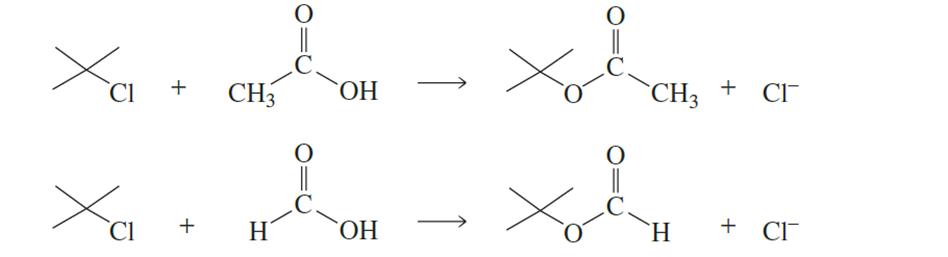
Which reactant in each of the following pairs undergoes an elimination reaction more rapidly? Explain your choice.


What do you think about this solution?
We value your feedback to improve our textbook solutions.