Chapter 9: Q88P (page 453)
Which reactant in each of the following pairs undergoes an elimination reaction more rapidly? Explain your choice.

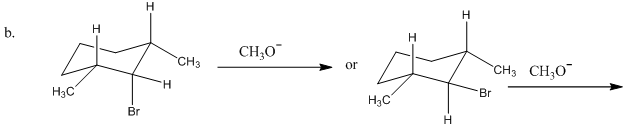
Short Answer
- (CH3)3CBr
Learning Materials
Features
Discover
Chapter 9: Q88P (page 453)
Which reactant in each of the following pairs undergoes an elimination reaction more rapidly? Explain your choice.



All the tools & learning materials you need for study success - in one app.
Get started for free
Which species in each pair is more stable?

a. Explain why the reaction of an alkyl halide with ammonia gives a low yield of primary amine.
b. Explain why a much better yield of primary amine is obtained from the reaction of an alkyl halide with an azide ion (-N3), followed by catalytic hydrogenation. (Hint: An alkyl azide is not nucleophilic.)
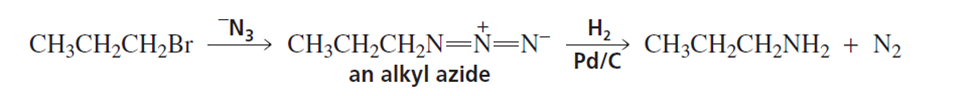
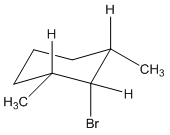
What products are formed when the following stereoisomer of 2-chloro-1,3-dimethylcyclohexane reacts with methoxide ion?

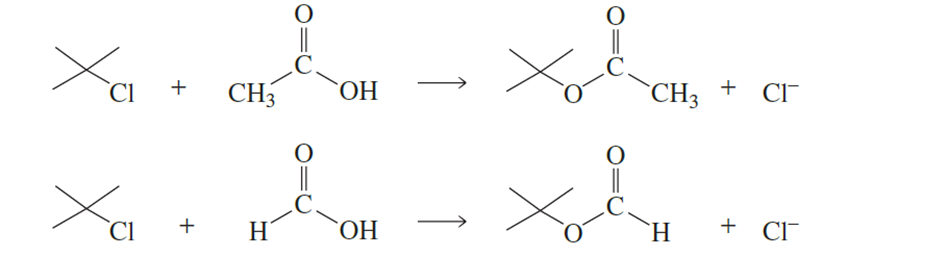
tert-Butyl chloride undergoes solvolysis in both acetic acid and formic acid. Solvolysis occurs 5000 times faster in one of these two solvents than in
the other. In which solvent is solvolysis faster? Explain your answer. (Hint: Formic acid is more polar than acetic acid.)
Starting with cyclohexene, how could the following compounds be prepared?
a. methoxycyclohexane
b. cyclohexylmethylamine
c. dicyclohexyl ether
What do you think about this solution?
We value your feedback to improve our textbook solutions.