Chapter 9: Q125P (page 391)
What products are formed when the following stereoisomer of 2-chloro-1,3-dimethylcyclohexane reacts with methoxide ion?
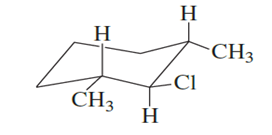
Short Answer
The products formed by Substitution and elimination reactions are given as below,

Learning Materials
Features
Discover
Chapter 9: Q125P (page 391)
What products are formed when the following stereoisomer of 2-chloro-1,3-dimethylcyclohexane reacts with methoxide ion?

The products formed by Substitution and elimination reactions are given as below,

All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the substitution products for each of the following reactions; if the products can exist as stereoisomers, show what stereoisomers are obtained:
a. ( R )-2-bromopentane + CH3O-
b. ( R )-3-bromo-3-methylheptane + CH3OH
c. benzyl chloride + CH3CH2OH
d. allyl chloride + CH3OH
e. 1-bromo-2-butene + CH3O-
f. 1-bromo-2-butene + CH3OH
Draw the major product obtained when each of the following alkyl halides undergoes an E2 reaction:
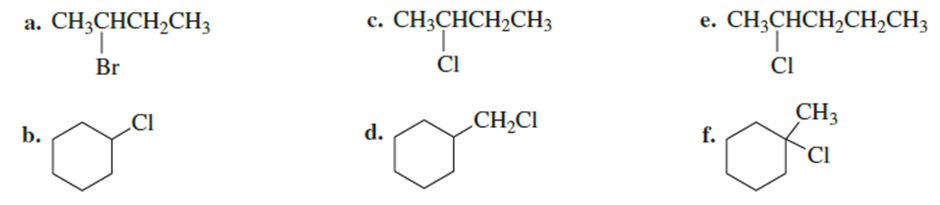
Indicate which species in each pair gives a higher substitution-product-to-elimination-product ratio when it reacts with isopropyl bromide:
a. ethoxide ion or tert-butoxide ion
b. -OCN or -SCN
c. Cl- or Br-
d. CH3S- or CH3O-
Draw the products obtained from the solvolysis of each of the following compounds in ethanol:
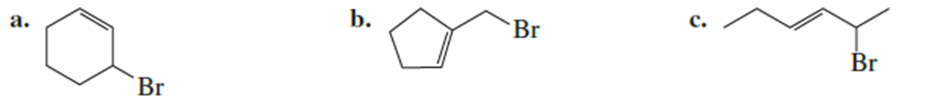
Draw the structure of DDE.
What do you think about this solution?
We value your feedback to improve our textbook solutions.