Chapter 9: Q86P (page 452)
Which species in each pair is more stable?

Short Answer
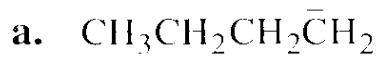


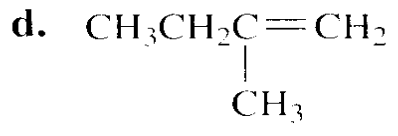
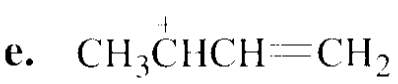
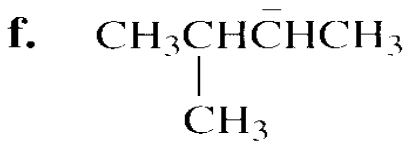
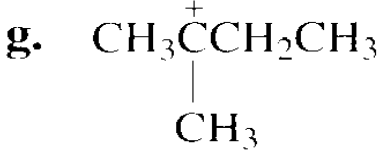
Learning Materials
Features
Discover
Chapter 9: Q86P (page 452)
Which species in each pair is more stable?



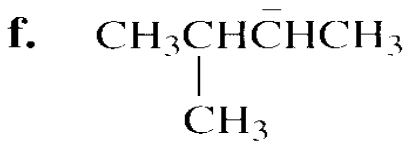
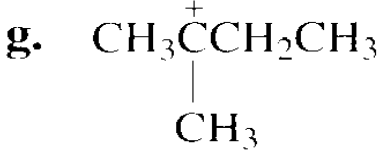
All the tools & learning materials you need for study success - in one app.
Get started for free
Four alkenes are formed from the E1 reaction of 3-bromo-2,3-dimethylpentane and methoxide ion. Draw the structures of the alkenes and rank them according to the amount that would be formed.
Question:Draw the elimination products for each of the following E2 reactions; if the products can exist as stereoisomers, indicate which stereoisomers are obtained.
a. (2S,3S)-2-chloro-3-methylpentane + high concentration of CH3O-
b.(2S,3R)-2-chloro-3-methylpentane + high concentration of CH3O-
c.(2R,3S)-2-chloro-3-methylpentane + high concentration of CH3O-
d.(2R,3R)-2-chloro-3-methylpentane + high concentration of CH3O-
e.3-chloro-3-ethyl-2,2-dimethylpentane + high concentration of CH3CH2O-
Draw the substitution products for each of the following reactions; if the products can exist as stereoisomers, show what stereoisomers are obtained:
a. ( R )-2-bromopentane + CH3O-
b. ( R )-3-bromo-3-methylheptane + CH3OH
c. benzyl chloride + CH3CH2OH
d. allyl chloride + CH3OH
e. 1-bromo-2-butene + CH3O-
f. 1-bromo-2-butene + CH3OH
Amines are good nucleophiles, even though they are neutral. How would the rate of an SN2 reaction between an amine and an alkyl halide be affected if the polarity of the solvent is increased?
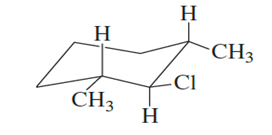
What products are formed when the following stereoisomer of 2-chloro-1,3-dimethylcyclohexane reacts with methoxide ion?
What do you think about this solution?
We value your feedback to improve our textbook solutions.