Chapter 9: Q45P (page 435)
What products will be obtained from the E2 reaction of the following alkyl halides?
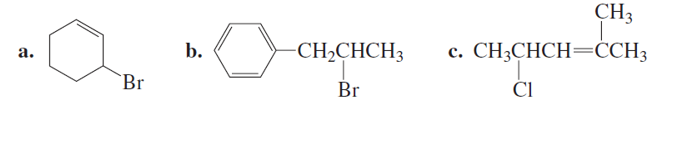
Short Answer
The products formed from the E2 mechanism are given below,
Learning Materials
Features
Discover
Chapter 9: Q45P (page 435)
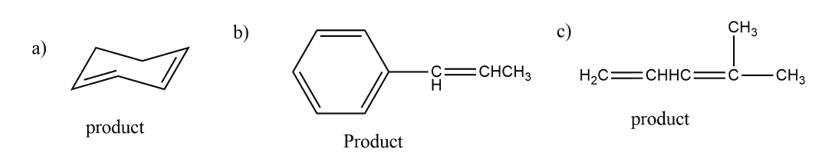
What products will be obtained from the E2 reaction of the following alkyl halides?

The products formed from the E2 mechanism are given below,

All the tools & learning materials you need for study success - in one app.
Get started for free
Which is a better nucleophile?
a. Draw the structures of the products obtained from the reaction of each enantiomer of cis-1-chloro-2-isopropylcyclopentane with sodium methoxide in methanol.
b. Are all the products optically active?
c. How would the products differ if the starting material were the trans isomer? Are these products optically active?
d. Will the cis enantiomers or the trans enantiomers form substitution products more rapidly?
e. Will the cis enantiomers or the trans enantiomers form elimination products more rapidly?
Why do cis-1-bromo-2-ethylcyclohexane and trans-1-bromo-2-ethylcyclohexane form different major products when they undergo an E2 reaction?
Draw the elimination products that are formed when 3-bromo-3-methyl-1-butene reacts with
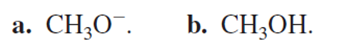
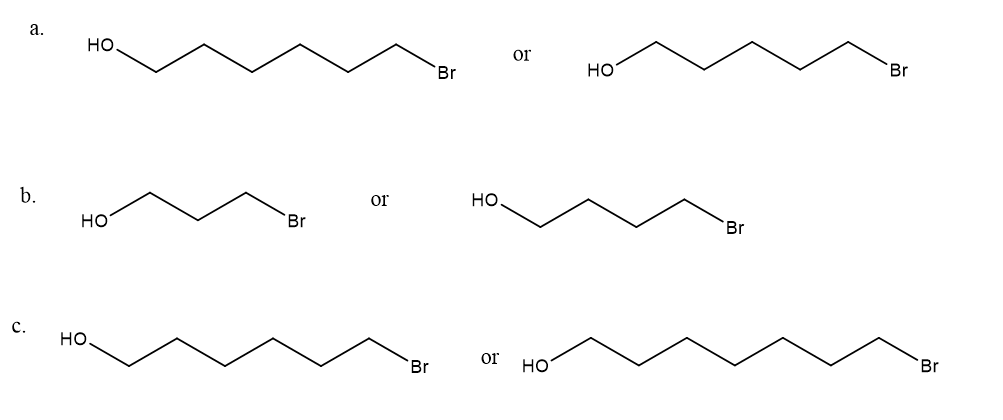
After a proton is removed from the OH group, which compound in each pair forms a cyclic ether more rapidly?
What do you think about this solution?
We value your feedback to improve our textbook solutions.