Chapter 9: Q25P (page 419)
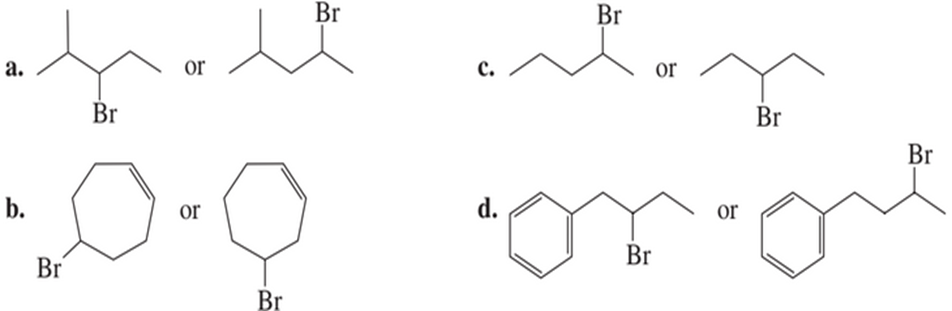
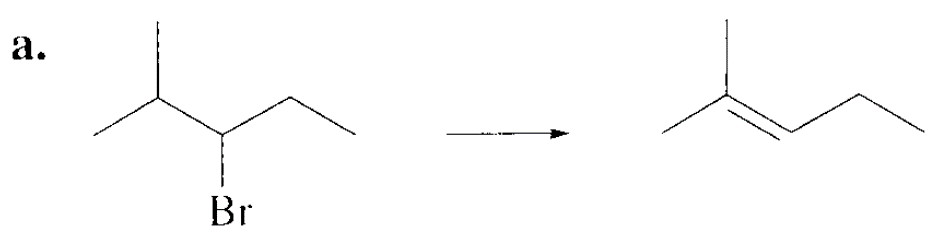
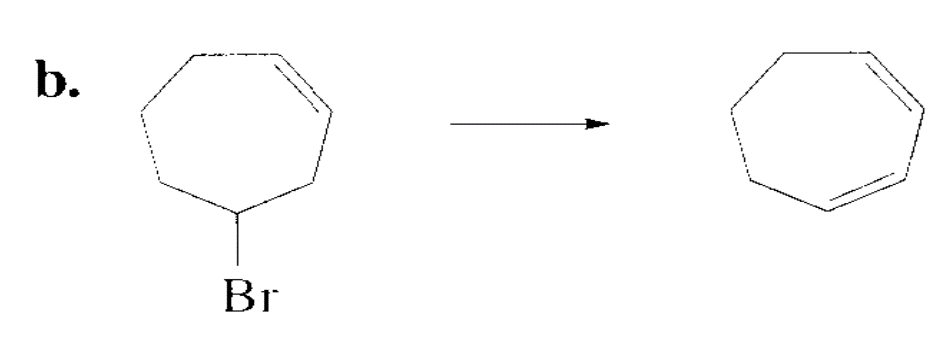
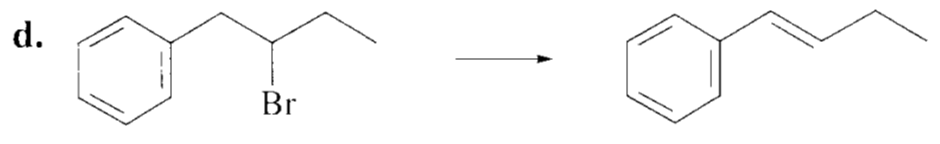
Which alkyl halide in each pair is more reactive in an E2 reaction with hydroxide ions?
Short Answer

Learning Materials
Features
Discover
Chapter 9: Q25P (page 419)
Which alkyl halide in each pair is more reactive in an E2 reaction with hydroxide ions?





All the tools & learning materials you need for study success - in one app.
Get started for free
Four alkenes are formed from the E1 reaction of 3-bromo-2,3-dimethylpentane and methoxide ion. Draw the structures of the alkenes and rank them according to the amount that would be formed.
Rank the following species in each set from best nucleophile to poorest nucleophile.

a. Identify the substitution products that form when 2-bromo-2-methylpropane is dissolved in a mixture of 80% ethanol and 20% water.
b. Explain why the same products are obtained when 2-chloro-2-methylpropane is dissolved in a mixture of 80% ethanol and 20% water.
Methoxychlor is an insecticide that was intended to take DDT’s place because it is not as soluble in fatty tissues and is more readily biodegradable. It, too, can accumulate in the environment, however, so its use was also banned—in 2002 in the European Union and in 2003 in the United States. Why is methoxychlor less soluble in fatty tissues than DDT?
What is the major elimination product obtained from the reaction of each of the following alkyl halides with hydroxide ions?

What do you think about this solution?
We value your feedback to improve our textbook solutions.