Chapter 18: Q93P (page 921)
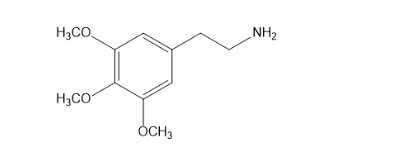
Describe how mescaline can be synthesized from benzene. The structure of mescaline is given on page 869.
Short Answer
.
Learning Materials
Features
Discover
Chapter 18: Q93P (page 921)
Describe how mescaline can be synthesized from benzene. The structure of mescaline is given on page 869.

.
All the tools & learning materials you need for study success - in one app.
Get started for free
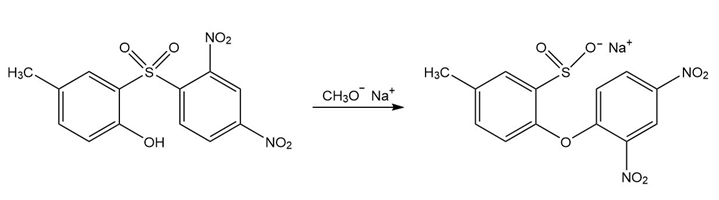
Propose a mechanism for each of the following reactions:
a.
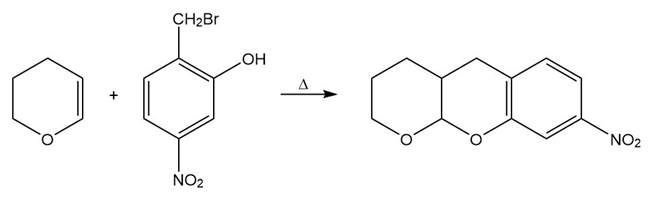
b.
Show how the following compounds can be synthesized from benzene:
a. N,N,N-trimethylanilinium iodide
b. 2-methyl-4-nitrophenol
c. p-benzylchlorobenzene
d. benzyl methyl ether
e. p-nitroaniline
f. m-bromoiodobenzene
g. p-dideuteriobenzene
h. p-nitro-N-methylaniline
i. 1-bromo-3-nitrobenzene
Show how each of the following compounds can be synthesized from benzene:
a. o-nitrophenol
b. p-nitroaniline
c. p-bromoanisole
d. anisole
Draw the structure of the activated benzene ring and the diazonium ion used in the synthesis of each of the following compounds, whose structures can be found on page 607.
a. butter yellow
b. methyl orange
a. Rank the following esters from most reactive to least reactive in the first slow step of a nucleophilic acyl substitution reaction (formation of the tetrahedral intermediate):
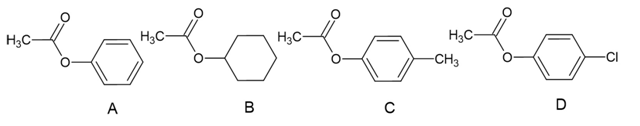
b. Rank the same esters from most reactive to least reactive in the second slow step of a nucleophilic acyl substitution reaction (collapse of the tetrahedral intermediate).
What do you think about this solution?
We value your feedback to improve our textbook solutions.