Chapter 18: Q94P (page 921)
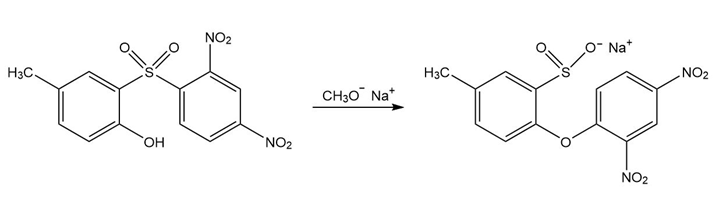
Propose a mechanism for the following reaction that explains why the configuration of the asymmetric center in the reactant is retained in the product:
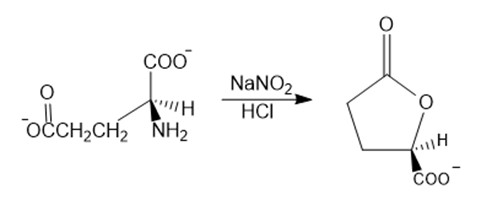
Short Answer
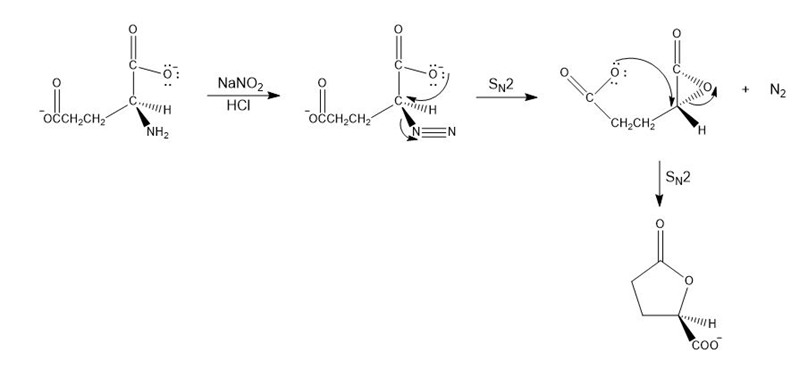
Mechanism of the given reaction
Learning Materials
Features
Discover
Chapter 18: Q94P (page 921)
Propose a mechanism for the following reaction that explains why the configuration of the asymmetric center in the reactant is retained in the product:


Mechanism of the given reaction
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the product(s) of each of the following reactions:
Friedel–Crafts alkylations can be carried out with carbocations formed from reactions other than the reaction of an alkyl halide with AlCl3. Propose amechanism for the following reaction:
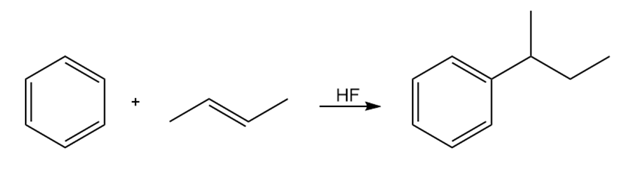
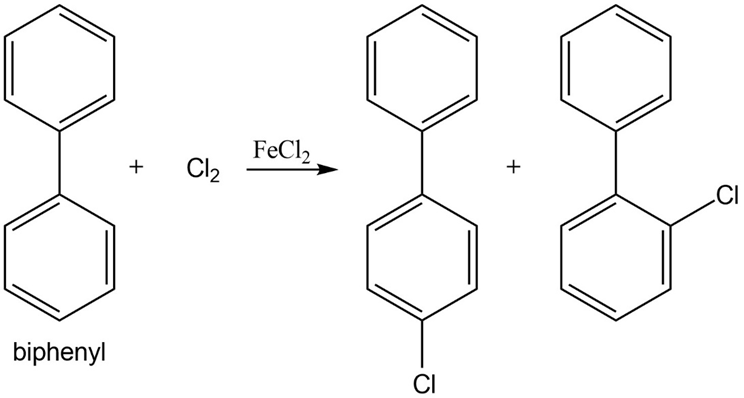
Propose a mechanism for each of the following reactions:
a.
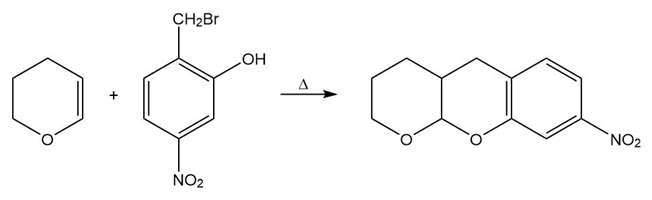
b.
Show how Novocain, a painkiller used frequently by dentists, can be prepared from benzene and compounds containing no more than four carbons.
 Novocain
Novocain
Using resonance contributors for the carbocation intermediate, explain why a phenyl group is an ortho–para director.
What do you think about this solution?
We value your feedback to improve our textbook solutions.