Chapter 18: Q95P (page 921)
Propose a mechanism for each of the following reactions:
a.
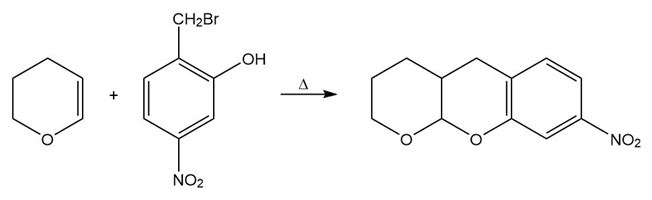
b.
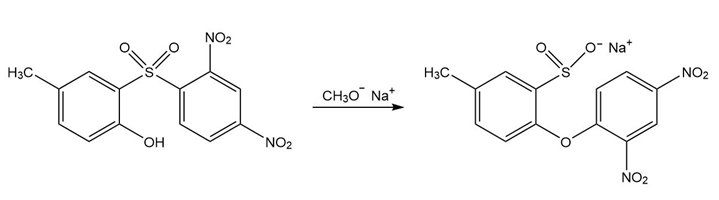
Short Answer
a.
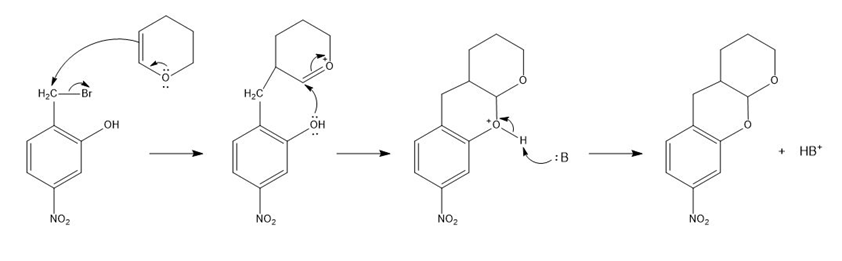
Mechanism for reaction (a)
b.
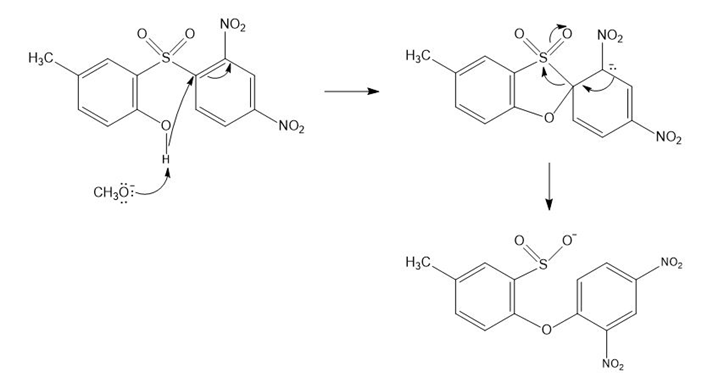
Mechanism for reaction (b)
Learning Materials
Features
Discover
Chapter 18: Q95P (page 921)
Propose a mechanism for each of the following reactions:
a.

b.

a.

Mechanism for reaction (a)
b.

Mechanism for reaction (b)
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw resonance contributors for the carbanion that would be formed if meta-chloronitrobenzene were to react with hydroxide ion. Why doesn’t the reaction occur?
What product is formed from the reaction of p-methylphenol with benzenediazonium chloride?
A mixture of 0.10 mol benzene and 0.10 mol p-xylene was allowed to react with 0.10 mol nitronium ion until all the nitronium ion was gone. Two products were obtained: 0.002 mol of one and 0.098 mol of the other.
Draw resonance contributors for the carbanion that would be formed if meta-chloronitrobenzene were to react with hydroxide ion. Why doesn’t the reaction occur?
Write the sequence of steps required for the conversion of benzene into benzenediazonium chloride.
What do you think about this solution?
We value your feedback to improve our textbook solutions.