Chapter 18: Q40P (page 908)
Show how each of the following compounds can be synthesized from benzene:
a. o-nitrophenol
b. p-nitroaniline
c. p-bromoanisole
d. anisole
Short Answer
a.
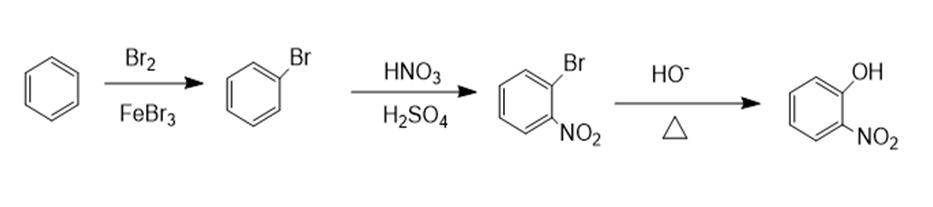
b.
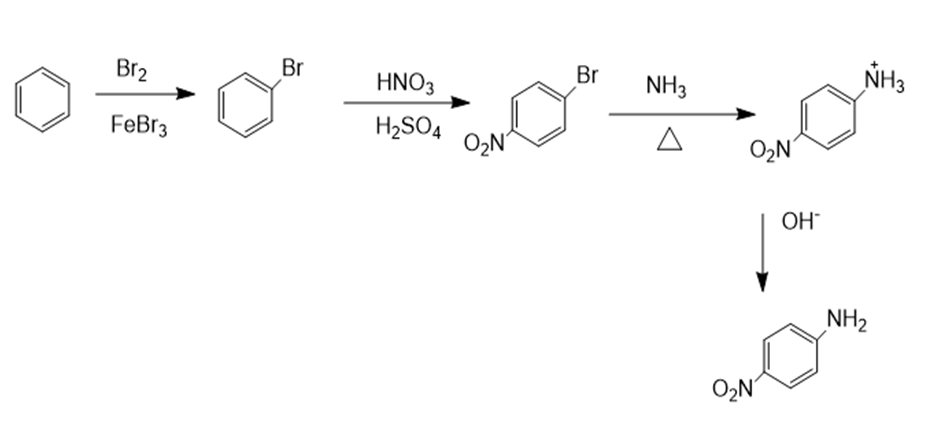
c.
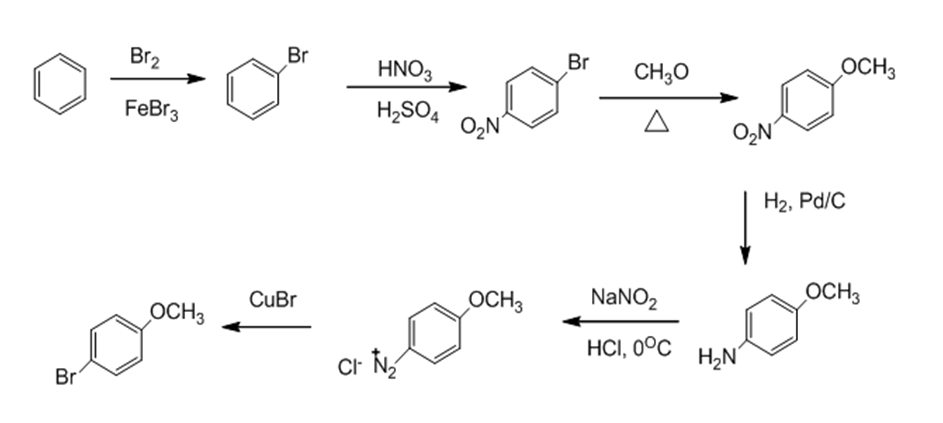
d.
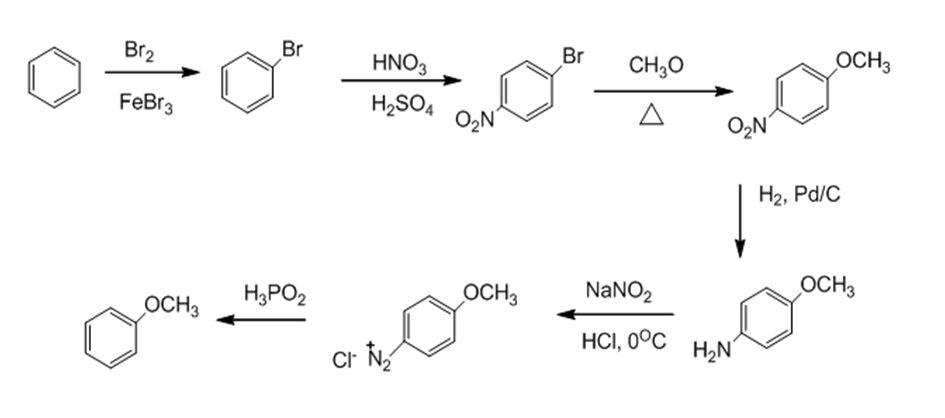
Learning Materials
Features
Discover
Chapter 18: Q40P (page 908)
Show how each of the following compounds can be synthesized from benzene:
a. o-nitrophenol
b. p-nitroaniline
c. p-bromoanisole
d. anisole
a.

b.

c.

d.

All the tools & learning materials you need for study success - in one app.
Get started for free
Explain why fluorobenzene is more reactive than chlorobenzene toward electrophilic aromatic substitution but chloromethyl benzene is more reactive than fluoromethylbenzene.
Describe two ways to prepare anisole from benzene.
a.Describe three ways the following reaction can be carried out:
b.Describe two ways the following reaction can be carried out:
An aromatic hydrocarbon with a molecular formula of C13H20has an 1H NMR spectrum with a signal at ~7 ppm that integrates to 5H. It also has two singlets; one of the singlets has 1.5 times the area of the second. What is the structure of the aromatic hydrocarbon?
If anisole is allowed to sit in that contains a small amount of, what products are formed?
What do you think about this solution?
We value your feedback to improve our textbook solutions.